1. INTRODUCTION
1.1. Background of Wound Healing and Global Health Burden
Wound healing is a fundamental biological process essential for maintaining tissue integrity and restoring barrier function following injury. This intricate process unfolds through a precisely orchestrated cascade of overlapping phases: hemostasis, inflammation, proliferation, and remodeling (Ramalingam et al., 2022). The initial response involves hemostasis, where vasoconstriction and platelet aggregation form a fibrin clot, providing a temporary matrix for subsequent cellular infiltration and a reservoir of growth factors (Rodrigues et al., 2019). This is followed by the inflammatory phase, characterized by the recruitment of neutrophils and macrophages to clear debris and pathogens, releasing pro-inflammatory cytokines that prepare the wound for new tissue formation (Ramalingam et al., 2022). The proliferative phase then ensues, involving fibroblast proliferation and migration, granulation tissue formation, angiogenesis, and reepithelialization by keratinocytes (Ramalingam et al., 2022). The remodeling phase involves collagen maturation and reorganization, leading to scar tissue formation (Ramalingam et al., 2022).
In spite of the body’s remarkable capacity for self-repair, disruptions in this delicate sequence, particularly because of underlying comorbidities, such as diabetes and obesity, frequently lead to chronic, nonhealing wounds (Zawani & Fauzi, 2021). These persistent wounds represent a substantial global health burden, significantly impairing patient quality of life and imposing immense economic strain on healthcare systems worldwide (Han & Ceilley, 2017). The majority of wounds in the global population are surgical or traumatic wounds (48.00%), leg ulcers (28.00%), and decubitus wounds (21.00%). A wound care association in United States conducted a study on the global incidence of wounds based on disease etiology and reported data on surgical wounds. Of the 110.30 million cases annually, 1.60 million cases were trauma wounds, 20.40 million cases were abrasions, 10 million cases were burns, 8.50 million cases were decubitus ulcers, 12.50 million cases were venous ulcers, 13.50 million cases were diabetic ulcers, 0.20 million cases were amputations, 0.60 million cases were carcinoma, 0.10 million cases were melanoma, and 0.10 million cases were complications related to skin cancer. For instance, chronic wounds collectively cost the United States healthcare system over US$25 billion annually, with a single diabetic ulcer potentially incurring costs of nearly US$50,000 (Han & Ceilley, 2017). The increasing global prevalence of diabetes and obesity further contributes to the escalating number of individuals affected by chronic wounds (Han & Ceilley, 2017). The disruption of healthcare services during the COVID-19 pandemic further compounded this burden, leading to reduced compliance with wound care visits and encouraging less effective, alternative management strategies (Sen, 2021). The economic consequences are profound, as delays in wound diagnosis and treatment directly correlate with increased hospitalization rates and higher acute care service utilization (Gould & Herman, 2025). The persistent challenge posed by chronic wounds highlights a critical gap in current healthcare practices. In spite of the significant economic and social impact, the field of wound care remains under-resourced, often overlooked, and inadequately represented in modern professional medical training curricula (Gould & Herman, 2025). This deficiency in foundational wound education and management among primary care clinicians contributes to diagnostic delays and inconsistent treatment standards (Gould & Herman, 2025). The complex nature of wound healing, coupled with a lack of comprehensive understanding, creates a scenario where a substantial public health issue is not adequately addressed. Addressing this global health burden necessitates not only continued scientific exploration for novel therapies but also fundamental reforms in medical education and healthcare infrastructure to promote early diagnosis, standardized management protocols, and the effective deployment of interprofessional care teams (Gould & Herman, 2025). The substantial financial drain underscores the urgency of these systemic improvements, as optimized wound care can significantly reduce hospitalizations and associated costs (Gould & Herman, 2025).
1.2. Limitations of Conventional Wound Therapies
Conventional wound care, in spite of ongoing advancements, faces persistent limitations—especially in chronic cases. These span across topical antimicrobials, dressings, skin substitutes, and growth factor therapies. Diagnostic tools like surface cultures offer limited accuracy, while deep tissue sampling may worsen the wound (Han & Ceilley, 2017). Common agents such as silver sulfadiazine lack strong evidence for healing efficacy, and iodine-based compounds raise toxicity concerns when used extensively (Han & Ceilley, 2017). Indiscriminate antibiotic use further drives antimicrobial resistance, including Methicillin-resistant Staphylococcus aureus (MRSA), complicating recovery (Han & Ceilley, 2017). Modern dressings also have drawbacks. Hydrocolloids are unsuitable for wounds with heavy exudate, while alginates may harm dry wounds (Han & Ceilley, 2017). Although antimicrobial dressings help manage biofilms, their broad use isn’t always suitable (Han & Ceilley, 2017). Advanced options like skin substitutes are costly, with limited proof of general effectiveness and potential risks of rejection or hypersensitivity (Han & Ceilley, 2017).
Growth factor therapies, in spite of their targeted biological mechanisms, have shown limited clinical success; for instance, platelet-derived growth factor (PDGF) has yielded only modest improvements in wound healing outcomes (Han & Ceilley, 2017). This limited efficacy is largely attributed to the molecular complexity of chronic wounds, which involve disruptions across numerous growth factors and cytokines—making single-agent supplementation insufficient to restore normal healing dynamics (Han & Ceilley, 2017). High development costs and difficulties in interpreting data from diverse wound models further hinder the widespread adoption of these therapies (Han & Ceilley, 2017). Even specialized interventions like hyperbaric oxygen therapy face scrutiny; although theoretically beneficial for fibroblast proliferation, immune modulation, and angiogenesis, clinical results remain inconsistent, and the systemic nature of the treatment introduces risks such as myopia, oxygen toxicity, and pneumothorax—restricting its use to specific cases like ischemic diabetic ulcers (Han & Ceilley, 2017). Conventional wound care also grapples with bacterial contamination and biofilm formation, often necessitating prolonged antibiotic use that contributes to antimicrobial resistance and diminishes treatment efficacy (Han & Ceilley, 2017). These limitations underscore the need for multitargeted, cost-effective therapeutic strategies capable of addressing the multifactorial pathology of chronic wounds.
1.3. The Potential of Plant-Based Bioactives in Wound Management
The growing limitations of conventional wound therapies have prompted a shift toward complementary and alternative medicines (CAMs), particularly plant-based bioactives (Ramalingam et al., 2022). Botanicals, long used in traditional medicine, are gaining scientific attention for their favorable safety profiles, lower production costs, and suitability for managing chronic wounds (Ramalingam et al., 2022). These compounds promote healing through antimicrobial, anti-inflammatory, angiogenic, and antioxidant mechanisms, while also enhancing cell proliferation (Quave, 2018). Their pharmacological diversity stems from a rich array of secondary metabolites—flavonoids, alkaloids, saponins, and phenolics—that enable interaction with multiple biological targets, offering a holistic alternative to single-target synthetic drugs (Quave, 2018). Among these, green tea is especially notable for its polyphenol-rich extracts, including catechins such as EGCG, EGC, ECG, and EC, which exhibit well-documented wound healing properties (Rodrigues et al., 2019). The continued exploration of such bioactives holds promise for developing effective and accessible wound care solutions
1.4. Objective of the Review
The primary objective of this comprehensive literature review is to systematically evaluate the current scientific evidence pertaining to the role of green tea extract (GTE) in wound healing. This evaluation will encompass a thorough analysis of findings derived from various research methodologies, specifically including in silico, in vitro, in vivo (animal models), and clinical studies. By synthesizing data across these diverse research levels, this review aims to provide a holistic understanding of GTE’s efficacy, its underlying mechanisms of action, and its potential as a therapeutic agent in wound care. Furthermore, the review will identify existing challenges and limitations in the current body of research and propose critical future directions to facilitate the translation of promising preclinical findings into robust clinical applications.
2. PHYTOCHEMISTRY AND PHARMACOLOGICAL PROPERTIES OF GREEN TEA
Green tea is renowned for its diverse health benefits, largely attributed to its rich phytochemical composition. The primary components of green tea are polyphenols, which constitute a significant proportion, ranging from 25% to 35% of the dry weight of the tea leaves (Gould & Herman, 2025). These polyphenols are critical determinants of the tea’s taste, particularly its characteristic bitterness and astringency (Khan et al., 2025).
2.1. Major Bioactive Compounds: EGCG, EGC, ECG, EC, Catechins, and Polyphenols
Catechins are the most prominent polyphenols in green tea, comprising about 70% of its total polyphenolic content and 30–42% of the leaf’s dry weight (Gould & Herman, 2025). As members of the flavan-3-ol subclass of flavonoids (Khan et al., 2025), they include EGCG—the most abundant and bioactive—making up 50–70% of total catechins, followed by EGC (≈19%), ECG (≈13.6%), and EC (≈6.4%) (Gould & Herman, 2025). Minor catechins like catechin and gallocatechin are also present (Khan et al., 2025). Green tea additionally contains flavonoids, anthocyanins, and phenolic acids, with polyphenols comprising up to 19.18% of dry leaf weight (Gould & Herman, 2025). These compounds act as hydroxyl radical scavengers, protecting cellular components such as lipids, proteins, and DNA from oxidative damage (Gould & Herman, 2025). Flavonols like myricetin, quercetin, and behenyl glycosides contribute 2–3% of the tea infusion, while phenolic acids such as chlorogenic, gallic, and caffeic acids, though less concentrated, enhance its bioactivity (Khan et al., 2025). Catechin levels vary based on leaf maturity, plant variety, geography, and processing methods—juvenile leaves contain more EGCG and ECG, whereas older leaves are richer in EGC and EC (Khan et al., 2025). Preparation techniques like steaming versus pan-firing also influence catechin composition (Khan et al., 2025).
2.2. Mechanisms of Action: Antioxidant, Anti-inflammatory, Antimicrobial, Angiogenic, and Collagen-Promoting
The diverse health benefits of green tea, particularly in wound healing, are mediated by the multifaceted pharmacological actions of its bioactive compounds.
2.2.1. Antioxidant Activity
EGCG, the most abundant catechin, functions as a potent antioxidant, primarily because of its phenolic groups that are susceptible to oxidation and can generate quinone structures (Singh et al., 2011). It effectively scavenges ROS such as superoxide, hydrogen peroxide (H2O2), and hydroxyl radicals, and can chelate transition metals, thereby preventing oxidative damage in healthy cells (Forester & Lambert, 2011). EGCG also enhances cellular antioxidant defense mechanisms by increasing the levels of endogenous antioxidant enzymes, including superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GSH-Px) (Sun et al., 2024, Azis et al., 2025). This is often mediated through the activation of the nuclear factor erythroid 2-related factor 2 (Nrf2) pathway, a key transcription factor that upregulates the expression of these protective enzymes (Sun et al., 2024). While primarily an antioxidant, green tea polyphenols (GTPs) can also act as pro-oxidants in vitro and in vivo by generating H2O2, a property whose role in therapeutic effects is still being elucidated (Forester & Lambert, 2011).
2.2.2. Anti-inflammatory Effects
Green tea catechins, especially EGCG and ECG, show strong anti-inflammatory effects by modulating key signaling pathways (Viaña-Mendieta et al., 2022). EGCG inhibits NF-κB activation by blocking IκB-α phosphorylation and suppressing IKK activity (Singh et al., 2010; 2011). These polyphenols reduce pro-inflammatory cytokines like TNF-α, IL-1β, IL-6, IL-8, and IL-17, while boosting anti-inflammatory cytokines IL-4 and IL-10 (Oyetakin-White et al., 2012). EGCG also inhibits COX-2, helping resolve chronic inflammation and supporting wound healing (Krzyszczyk et al., 2018; Singh et al., 2011). Studies confirm that GTPs suppress inflammation through multiple mechanisms (Gould & Herman, 2025). They inhibit cytokine expression via NF-κB suppression (Sen, 2021), reduce COX-2 and iNOS activity to lower prostaglandin and nitric oxide levels (Quave, 2018), and activate the Nrf2/HO-1 pathway to enhance antioxidant defenses. Green tea also modulates immune responses by decreasing IL-17 and increasing IL-10 (Quave, 2018).
2.2.3. Antimicrobial Activity
Green tea extracts (GTE) and EGCG possess notable antimicrobial properties against a wide range of wound-related pathogens. Studies have demonstrated their ability to inhibit the growth of various Gram-positive bacteria, including Staphylococcus aureus and MRSA, as well as Gram-negative bacteria such as Pseudomonas aeruginosa and Escherichia coli (Taylor et al., 2005). The mechanisms underlying this activity are diverse and include direct binding to bacterial cell membranes, leading to increased permeability, H2O2 generation, and subsequent membrane damage, which can prevent bacterial attachment to host cells (Khan et al., 2025). Catechins can also inhibit key bacterial enzymes, such as dihydrofolate reductase (DHFR), and interfere with essential biosynthetic pathways like fatty acid biosynthesis (Reygaert, 2018). Furthermore, EGCG has been shown to inhibit biofilm formation and quorum sensing in pathogens like P. aeruginosa, which are critical virulence factors in chronic wound infections (Reygaert, 2018). Notably, green tea catechins can exhibit synergistic effects when combined with conventional antibiotics, such as gentamicin, enhancing their antimicrobial action against resistant strains (Bazzaz et al., 2016).
2.2.4. Angiogenic Properties
In the context of wound healing, GTE and its components have been shown to promote angiogenesis, the formation of new blood vessels from preexisting vasculature (Rodrigues et al., 2019). Studies have shown that GTE can increase neovascularization and promote the formation of organized granulation tissue (Safoine et al., 2024). While some studies, particularly in cancer research, indicate that EGCG can inhibit vascular endothelial growth factor (VEGF) and matrix metalloproteinase-9 (MMP-9) signaling and angiogenesis (Leong et al., 2009), the context of wound healing often requires the promotion of new vessel formation. The overall evidence in wound healing models suggests a beneficial role for green tea in promoting angiogenesis, likely through complex modulation of various growth factors and signaling pathways (Leong et al., 2009). In addition, green tea inhibits the activation of signal transducer and activator of transcription (STAT3), a transcription factor that regulates VEGF and MMP-9 expression, thereby limiting angiogenesis in cancer cells (Taylor et al., 2005). EGCG also modulates the phosphatidylinositol-3-kinase/Protein kinase B (PI3K/AKT) and mitogen-activated protein kinase (MAPK) signaling pathways, which are involved in endothelial cell proliferation and migration (Jeon et al., 2014).
2.2.5. Collagen-Promoting Effects
Green tea and its catechins play a significant role in promoting collagen synthesis and deposition, which are fundamental for wound closure and tissue remodeling (Zawani & Fauzi, 2021). Research indicates that EGCG can enhance the density of collagen and reticular fibers within connective tissues (Hajiaghaalipour et al., 2013). Histopathological evaluations of wounds treated with GTE often reveal increased collagen content and improved collagen organization, leading to reduced scar width (Hajiaghaalipour et al., 2013). EGCG has been shown to influence collagen type I gene regulation and modulate the activity of transforming growth factor-beta 1 (TGF-β1), a key factor in collagen production and fibroblast differentiation (Klass et al., 2010). Furthermore, EGCG can regulate MMPs, enzymes responsible for extracellular matrix (ECM) degradation. By reducing the expression of MMP-1 and MMP-2, EGCG helps prevent excessive collagen breakdown, which can otherwise impair wound healing (Fatemi et al., 2014). While EGCG can suppress the proliferation and migration of fibroblasts, particularly in pathological conditions like keloids, its overall effect in normal wound healing contributes to healthy granulation tissue formation and scar quality (Park et al., 2008).
2.3. Bioavailability and Stability Considerations of GTPs
In spite of the promising in vitro and in vivo pharmacological activities of GTPs, their clinical application is significantly influenced by considerations of bioavailability and stability. Bioavailability refers to the fraction of an ingested compound that reaches systemic circulation and the specific target tissues where it can exert its biological action (D’Archivio et al., 2010). In humans, the plasma bioavailability of green tea catechins, particularly EGCG and ECG, is generally low, with reported levels ranging from 0.07% to 0.20% after oral administration (D’Archivio et al., 2010). Even for EGC and EC, plasma content is relatively low (0.26–0.75%) (Khan & Mukhtar, 2013). However, preliminary reports indicate that catechins and their metabolites are indeed found in skin biopsies and blister fluid after green tea consumption, although concentrations are consistently higher in plasma than in skin (Clarke et al., 2016).
GTP bioavailability is influenced by factors such as chemical structure, concentration, food matrix composition, and processing methods like heating, homogenization, and storage (D’Archivio et al., 2010). Environmental conditions during plant growth and host-related factors, including enzyme activity and gut microbiota, also affect absorption and metabolism (D’Archivio et al., 2010). Gut microbes can convert catechins into aromatic acids, altering their bioactivity (Khan & Mukhtar, 2013). Catechin stability is another challenge. Temperature, pH, oxygen exposure, and metal ion interactions can degrade or alter their structure (Khan et al., 2025). Stabilizers like ascorbic acid and sugars help reduce oxidation and improve stability, though high doses of Vitamin C may accelerate degradation (Khan et al., 2025). In spite of these efforts, GTPs remain unstable and poorly bioavailable. After oral intake, catechins peak in blood within 1–2 hours and clear by 5–6 hours, requiring frequent dosing (Janle et al., 2008). However, EGCG may persist longer in tissue fluids, suggesting that sustained-release formulations may be less critical for tissue-level effects (Janle et al., 2008). In spite of these challenges, ongoing research into improved formulation and delivery systems aims to enhance the stability and bioavailability of these promising compounds (Table 1), which summarizes the key bioactive compounds of green tea and their primary pharmacological properties relevant to wound healing.
Table 1
Key bioactive compounds of green tea and their primary pharmacological properties relevant to wound healing.
3. IN SILICO STUDIES
In silico studies, leveraging computational methodologies, have become indispensable tools in modern drug discovery and mechanistic elucidation, particularly for complex natural products like GTE. These approaches enable the prediction of molecular interactions, pharmacokinetic profiles, and potential toxicities, thereby guiding subsequent in vitro and in vivo investigations.
3.1. Molecular Docking of EGCG and Other Catechins with Wound-Healing-Related Targets (e.g., MMPs, TGF-β, and VEGF)
Molecular docking is a computational technique used to predict the binding affinity and orientation of small molecules (ligands) with protein targets. This method is crucial for virtually screening bioactive compounds and simulating their interactions at a molecular level (Andrés et al., 2025). For green tea catechins, particularly EGCG, molecular docking studies have provided valuable insights into their potential targets involved in wound healing. The binding of EGCG and other catechins to various proteins is understood to be central to their diverse pharmacological actions (Nakano et al., 2018). The galloyl moiety of catechins, with its hydroxyl groups, is often identified as a key structural feature responsible for anchoring these compounds to protein clefts, explaining the higher activity of galloylated catechins like EGCG and epicatechin gallate (Nakano et al., 2018).
Key protein targets in wound healing explored through molecular docking with EGCG and other catechins include MMP-2 and MMP-9, zinc-dependent enzymes essential for ECM remodeling (Kandhwal et al., 2022). Dysregulated MMP activity can impair healing, and EGCG may regulate these enzymes to reduce cellular invasion and modulate scar formation (Fatemi et al., 2014; Negri et al., 2018). EGCG also interacts with TGF-β receptor II, potentially influencing fibroblast activity and collagen contraction, which may help prevent excessive scarring (Klass et al., 2010). VEGF, a key factor in angiogenesis, is another target. While EGCG may inhibit VEGF signaling in cancer models (Leong et al., 2009), its role in wound healing appears more supportive, with in vivo studies showing enhanced angiogenesis (Al-Rawaf et al., 2019; Eming et al., 2007). Other targets include Pin1, DNMTs, and HDACs, which regulate inflammation and epigenetic changes (Negri et al., 2018). EGCG’s interactions with these proteins reflect its broad effects on cell proliferation, survival, and immune responses (Negri et al., 2018).
3.2. Prediction of Pharmacokinetics and Toxicity
In silico methods also contribute to predicting the pharmacokinetic profile and potential toxicity of green tea catechins, which is crucial for rational drug design and clinical translation.
3.2.1. Pharmacokinetics
Preliminary computational and analytical reports indicate that green tea catechins and their metabolites are bioavailable in human skin. They have been detected in skin biopsies, blister fluid, and plasma after consumption (Clarke et al., 2016). However, the concentration of these catechins and their metabolites is consistently higher in plasma compared to the skin tissue (Clarke et al., 2016). Following oral consumption, green tea catechins typically reach peak blood concentrations within 1–2 hours and are eliminated from the systemic circulation within 5–6 hours (Janle et al., 2008). This rapid elimination suggests that frequent administration would be necessary to maintain sustained effective concentrations in the blood (Janle et al., 2008). Interestingly, studies on sustained-release GTE formulations indicate a broader distribution and longer duration of elevated catechin levels in interstitial fluid (tissue fluid) compared to plasma, suggesting that for tissue-specific effects, sustained-release might not always be critical (Janle et al., 2008). The metabolism of catechins by gut microbiota also impacts their final bioavailability and the forms present in the body (Clarke et al., 2016).
3.2.2. Toxicity Prediction
In silico models can aid in predicting potential toxicities. EGCG, while generally considered safe, has been shown to induce toxicity at high doses, with the severity being dependent on the dose, route of administration (e.g., intraperitoneal vs. oral), and duration of treatment (Ramachandran et al., 2016). Concerns regarding liver damage (hepatotoxicity) and potential drug interactions have been raised in some preclinical and clinical reports (Khan et al., 2025). For instance, EGCG can interact with drug transporters (e.g., P-glycoprotein) and drug-metabolizing enzymes, potentially altering the pharmacokinetics of co-administered medications (Khan et al., 2025). This highlights the importance of considering potential adverse effects and interactions, particularly with concentrated extracts or when consumed on an empty stomach, which can increase bioavailability and thus toxicity (Sarma et al., 2008).
3.3. Computational Pathway Analysis
Computational pathway analysis provides a systems-level understanding of how green tea bioactives interact with complex biological networks involved in wound healing. This approach identifies key biomolecular mechanisms and predicts quantitative effects on various cellular processes. One significant area of focus is the immunomodulatory effects of EGCG. Computational systems biology has identified that EGCG can enhance anti-inflammatory activity by upregulating the synthesis of HO-1, which is known to promote regulatory T cell (Treg) and T helper 2 (Th2) phenotypes associated with immune tolerance (Ayyadurai & Deonikar, 2021). Concurrently, EGCG downregulates the expression of several pro-inflammatory cytokines, including interleukin-2 (IL-2), IL-17, interferon-gamma (IFN-γ), TNF-α, nitric oxide (NO), IL-6, and IL-1β (Ayyadurai & Deonikar, 2021, Azis et al., 2025). This dual action of suppressing pro-inflammatory mediators while promoting anti-inflammatory pathways is crucial for resolving chronic inflammation in wounds.
Computational analysis also sheds light on the role of green tea in modulating microRNA (miRNA) expression. MiRNAs are small noncoding RNA molecules that play pivotal roles in regulating gene expression and biological processes, including wound healing (Al-Rawaf et al., 2019). Tissue oxygenation levels in wounds can affect the expression of specific miRNAs (oxymiRs). Studies have shown that GTE can influence the expression of hypoxia-responsive miRNAs (HRMs), such as miR-424, miR-199a, miR-210, and miR-21, which are critical for regulating angiogenesis and vascular remodeling in wound healing, particularly in diabetic wounds (Al-Rawaf et al., 2019). This suggests that green tea’s beneficial effects may, in part, be mediated through the regulation of these small RNA molecules. Computational approaches help identify how green tea bioactives affect key wound healing pathways, including PI3K/AKT, Wnt/β-catenin, TGF-β, Nrf2, and Notch (Bonnici et al., 2023). These pathways regulate cell proliferation, migration, angiogenesis, and macrophage polarization. In silico studies are essential for predicting molecular interactions and guiding the development of targeted wound therapies with improved efficacy and fewer side effects (Thomas et al., 2024). EGCG influences MMP signaling by inhibiting MMP-2 and MMP-9, aiding ECM remodeling and wound repair (Hao et al., 2021; Hajiaghaalipour et al., 2013). It also modulates TGF-β expression, which governs cell growth and migration (Fatemi et al., 2014). EGCG affects angiogenesis via AMPK activation, reducing VEGF and MMP-9 levels to regulate vascularization (Hajiaghaalipour et al., 2013). These signaling pathways interact with each other and are mediated by various regulatory proteins, as predicted through computational pathway analysis, providing a comprehensive understanding of the EGCG-mediated wound healing mechanism (Table 2), which summarizes key findings from in silico studies on GTE in wound healing.
Table 2 Summary of key findings from in silico studies on green tea extract in wound healing.
4. IN VITRO STUDIES
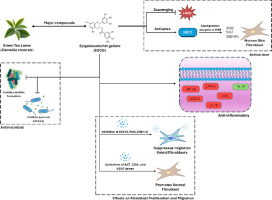
In vitro studies, conducted using cell cultures or isolated tissues, provide fundamental insights into the direct cellular and molecular effects of GTE and its components on various aspects of wound healing (Figure 1). These controlled experimental settings allow for the elucidation of specific mechanisms of action before progression to more complex in vivo models.
Fibroblasts play a pivotal role in wound healing, with their migration and proliferation being essential for granulation tissue formation (Rodrigues et al., 2019). EGCG has been shown to influence fibroblast proliferation and migration, inhibiting these processes more strongly in keloid fibroblasts compared to normal fibroblasts (IC50: 54.4 μM for keloid fibroblasts vs. 63.0 μM for normal fibroblasts) (Park et al., 2008), suggesting a potential role in modulating excessive scar formation. Although some studies on cancer cells, such as A549 lung cancer cells, demonstrated that EGCG suppresses proliferation and migration at concentrations close to or exceeding 50 μM (Kyriacou et al., 2025), other findings indicate that GTEs can promote fibroblast proliferation (Kumar et al., 2016). For instance, green tea and hyaluronic acid gels significantly enhanced expression of AKT, CDK, and VEGF genes, suggesting positive effects on human fibroblast proliferation (Bonatto et al., 2024). The scratch assay is commonly used to study migration (Balachandran et al., 2023), and EGCG shows context-dependent activity. It inhibits keloid fibroblast proliferation, suggesting anti-fibrotic and anti-scar potential (Park et al., 2008) but promotes normal fibroblast function to support repair (Kumar et al., 2016). Its stronger inhibition of keloid fibroblasts is linked to the suppression of STAT3, PI3K, and ERK1/2 pathways (Meng et al., 2020), underscoring EGCG’s dual role in treating fibrotic conditions while promoting balanced remodeling. In terms of inflammation, which is essential but detrimental when prolonged (Ramalingam et al., 2022), macrophages are central players that transition from M1-like to M2-like phenotypes, a shift often impaired in chronic wounds (Krzyszczyk et al., 2018). GTPs, particularly EGCG and ECG, modulate this process by inducing monocyte apoptosis (Forester & Lambert, 2011), suppressing mediators such as TNF-α, IL-1, IL-6, and IL-8 through NF-κB, AP-1, and MAPK inhibition (Oyetakin-White et al., 2012), and reducing histamine release from mast cells (Oyetakin-White et al., 2012). EGCG also decreases NO and ROS levels while inhibiting NF-κB signaling in LPS-stimulated RAW264.7 macrophages (Hossen et al., 2023), suppresses TNF-α and IL-6 in mast cells (Li et al., 2016), and enhances keratinocyte hydration by regulating filaggrin, HAS-1/2, and HYAL expression under UVB exposure (Ramalingam et al., 2022). In addition, it protects against oxidative stress–induced MAPK activation (Krzyszczyk et al., 2018), thereby reinforcing barrier function. Oxidative stress itself, driven by ROS and poor antioxidant defenses, impairs healing, but green tea catechins counteract this through radical scavenging and metal chelation (Forester & Lambert, 2011).
In vitro, EGCG alleviates H2O2-induced fibroblast damage by enhancing enzyme activity and reducing lipid peroxidation (Lian et al., 2014), while polycaprolactone/gelatin nanofibers delivering EGCG efficiently scavenge ROS in fibroblasts exposed to H2O2 or UV (Lian et al., 2014). Moreover, green tea polyphenols and caffeine inhibit ROS and 4-hydroxy-2-nonenal accumulation in stressed human fibroblasts (Farrar et al., 2015). Beyond cellular effects, bacterial contamination and biofilm formation are major barriers in chronic wounds (Soltani et al., 2014). GTE and EGCG exhibit strong antibacterial activity against Gram-positive and Gram-negative pathogens, including MRSA, P. aeruginosa, and E. coli (Jeon et al., 2014). EGCG’s MICs range from 200 to 400 µg/mL for P. aeruginosa and ~400 µg/mL for E. coli (Hao et al., 2021), acting through membrane disruption, H2O2 generation, and inhibition of enzymes such as DHFR and fatty acid biosynthesis (Jeon et al., 2014). It also impairs P. aeruginosa biofilms and quorum sensing by disrupting amyloid and cellulose fibrils (Hao et al., 2021). When combined with antibiotics like gentamicin, catechins enhance efficacy against resistant strains and reduce MIC values (Bazzaz et al., 2016). EGCG and ECG inhibit a broad bacterial spectrum and can reverse MRSA resistance to β-lactams, resensitizing S. aureus to oxacillin (Bazzaz et al., 2016). While GTE inhibits Micrococcus luteus and Bacillus cereus, S. aureus is less susceptible (Radji et al., 2013), and some studies report no effect on P. aeruginosa in disc diffusion assays (Radji et al., 2013), suggesting that outcomes depend on extract type and concentration. EGCG may therefore act more as a quorum sensing inhibitor than a direct bactericide (Avadhani et al., 2017), a mechanism particularly valuable against biofilm-forming pathogens (Table 3). This highlights the dual antimicrobial and anti-virulence actions of green tea, underscoring its potential as an adjuvant rather than a standalone antibiotic.
Table 3
Summary of in vitro studies on green tea/EGCG in wound healing.
| Source | Cell Type | Form of Green Tea/EGCG | Concentration/Dose | Observed Effect | Key Findings |
|---|---|---|---|---|---|
| (Park et al., 2008) | Keloid and normal fibroblasts | EGCG | IC50: 54.4 μM (keloid), 63.0 μM (normal) | More potent suppression of keloid fibroblast proliferation and migration | Inhibition of STAT3 signaling pathway |
| (Bobadilla et al., 2019) | Human fibroblasts | Green tea and hyaluronic acid gel | Not specified | Increased expression of AKT, CDK, and VEGF genes | Positive effect on cell proliferation and angiogenesis |
| (Bonatto et al., 2024) | A549 lung cancer cells | EGCG | ≥50 μM | Inhibition of cancer cell proliferation and migration | Reduced cell viability and restricted migration |
| (Leong et al., 2009) | RAW264.7 macrophages | EGCG | Not specified | Reduced cellular NO and intracellular ROS, inhibited P-IκB/IκB and p-p65/p65 ratios | NF-κB pathway inhibition |
| (Kim et al., 2018) | HaCaT keratinocytes | EGCG | Not specified | Increased expression of natural moisturizing factor–related genes, reduced HYAL expression | Promoted skin hydration and protection against oxidative stress |
| (Xu et al., 2021) | Human dermal fibroblasts | EGCG | Not specified | Increased SOD and GSH-Px activities, reduced MDA levels | Antioxidant activity, ROS scavenging |
| (Zawani & Fauzi, 2021) | Normal human skin fibroblasts (WS-1) | Green tea polyphenols and caffeine | 0.001% GTPs, 0.1 mM affeine | Inhibited H2O2-induced ROS and HNE elevation | Complementary antioxidant function |
| (Taylor et al., 2005) | Staphylococcus aureus (including MRSA) | EGCG, ECG | Sub-inhibitory | Reversed β-lactam antibiotic resistance | Disruption of bacterial cytoplasmic membrane function |
| (Chan et al., 2011) | Staphylococcus aureus, Micrococcus luteus, Bacillus cereus | Green tea extract | 1.0–2.0 mg/disc | Inhibited Gram-positive bacteria | Catechins identified as primary contributors |
5. IN VIVO STUDIES
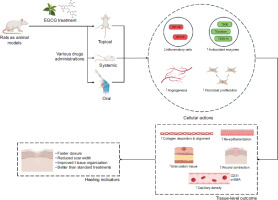
Various animal models have been employed to investigate the effects of green tea on wound healing, including full-thickness excisional wounds in rats, burn wounds in rats, and diabetic wound models in rats (Hosseini et al., 2020). The diabetic wound model is of particular significance because of the delayed healing challenges commonly observed in such patients (Tajammal et al., 2025). Topical administration of GTE on excisional wounds in rats has been shown to accelerate wound closure and reduce scar width (Saghazadeh et al., 2018). For instance, GTE (0.6% or 6% in a Vaseline base) applied topically for 14 days to excisional wounds in rats yielded improved healing outcomes (Karamchand et al., 2021), while 0.6% ethanol extract in Vaseline was applied for 21 days to surgical wounds in rats (Safoine et al., 2024). Collagen sponges containing EGCG (10 ppm) were also applied to full-thickness wounds in type 2 diabetic rats for 14 days, resulting in enhanced healing (Bonnici et al., 2023). Similarly, a 2% alcoholic extract of green tea was used as a first-aid treatment for 15 minutes daily on second-degree burn wounds in rats (Bonnici et al., 2023). In addition, various hydrogel or nanoparticle formulations containing EGCG have been tested, frequently demonstrating sustained release profiles and enhanced healing effects (Xu et al., 2021). Beyond topical use, daily oral administration of green tea catechin solution to rats for 10 days prior to sham laparotomy, followed by sacrifice on either day 7 or day 21 post-operation, has been reported (Hajiaghaalipour et al., 2013). Dietary GTE was also administered to rats for 3 weeks before spinal cord injury and maintained thereafter (Bonnici et al., 2023), while systemic EGCG administration in type 2 diabetic rats (specific daily dosage not reported) markedly accelerated wound healing (Radstake et al., 2023). Histopathological assessment remains a widely used method to evaluate microscopic changes in wound tissue, with parameters such as inflammatory cell infiltration, collagen deposition (quantity and organization), angiogenesis, and reepithelialization frequently analyzed (Soltani et al., 2014).
Consistently, wound contraction was significantly enhanced in green tea–treated groups, while reepithelialization was improved following treatment with GTE or EGCG, often resulting in earlier and more complete epithelial coverage (Gulua et al., 2024). Collagen and fibronectin deposition was also increased in green tea–treated wounds (Khan & Mukhtar, 2013), correlating with reduced scar width (Janle et al., 2008), and angiogenesis was promoted by green tea and EGCG, as evidenced by increased capillary density and elevated expression of markers such as Cluster of Differentiation 31 (CD31) (Saeki et al., 2018). Comparative studies with conventional wound therapies show that topical green tea was effective in treating second-degree burn wounds in rats, producing better outcomes than simple dressings, although not always achieving statistical significance compared to silver sulfadiazine or Vaseline (Saeki et al., 2018). Moreover, one study demonstrated that green tea dressings were as effective as conventional topical metronidazole powder in controlling odor in malodorous malignant wounds, suggesting its potential as an alternative to reduce the risk of drug resistance (Fukino et al., 2008) (Table 4). Although green tea exhibits positive effects in animal wound models, sometimes comparable to standard treatments, it does not consistently outperform established therapies across all parameters or models (Figure 2). Nevertheless, its comparable efficacy, natural origin, and potentially lower side-effect profile (Ohishi et al., 2016) position it as a strong candidate for adjunctive therapy, particularly in scenarios where conventional treatments are limited, such as drug resistance or cost constraints. This suggests that green tea’s role may be complementary rather than substitutive, especially in complex or chronic wounds where a multimodal therapeutic approach is advantageous, with its potential to reduce antimicrobial resistance (Hsu, 2005) representing a particularly valuable advantage.
Table 4
Summary of in vivo studies on green tea/EGCG in wound healing.
| Source | Animal Model | Wound Type | Form of Green Tea/EGCG & Concentration | Route of Administration | Treatment Duration | Key Outcomes | Histopathological Findings |
|---|---|---|---|---|---|---|---|
| (Hajiaghaalipour et al., 2013) | Sprague Dawley rats | Excision wound | Green tea extract (1 g leaves in 200 mL methanol) | Topical | 10 days | Accelerated wound healing, significant wound contraction, re-epithelialization | Reduced inflammatory cell infiltration, increased collagen deposition and angiogenesis, smaller scar width |
| (Shahein, 2017) | Sherman rats | Excisionwound | Green tea extract (0.6% and 6% in Vaseline) | Topical | 14 days | Significant increase in wound contraction rate and glutathione levels | Marked improvement in skin condition, granulation tissue formation, and epithelialization |
| (Asadi et al., 2013) | Wistar rats | Surgical wound | Green tea ethanol extract 0.6% in Vaseline | Topical | 21 days | Shorter healing duration (14.66 vs 20.66 days), greater wound length reduction | Significant differences in epithelial regeneration, granulation tissue thickness, and angiogenesis |
| (Chacko et al., 2010) | Wistar albino rats | Second-degree burn wound | Green tea alcohol extract 2% | Topical | 14 days | Significant reduction in burn wound size | Improved vascularization (not statistically significant) |
| (Kim et al., 2008) | Diabetic and non-diabetic rats | Excision wound | Green tea extract 0.6% | Topical | 3 weeks | Accelerated healing, greater collagen and fibronectin deposition, higher NO expression | Smaller scar width, enhanced re-epithelialization |
| (Fatemi et al., 2014) | STZ-induced diabetic rats | Skin wound | EGCG | Systemic (unspecified) | Not specified | Improved delayed wound healing, reduced macrophage accumulation, inhibited inflammatory response and Notch signaling | Not specifically reported |
| (Viaña-Mendieta et al., 2022) | Type 2 diabetic rats | Full-thickness wound | EGCG | Systemic | Daily | Strongly accelerated wound healing response, promoted re-epithelialization | Not specifically reported |
6. CLINICAL STUDIES
Clinical evidence for the role of green tea in wound healing remains limited, with most studies focusing on dermatological conditions such as UV-induced erythema, photoaging, or striae distensae rather than directly on acute or chronic wounds (Kumar et al., 2016). One preexperimental clinical study (n = 36 women) investigated a 3% GTE cream for striae distensae, applied for 8 weeks (Kumar et al., 2016), while a randomized, double-blind, controlled trial (n = 62 volunteers) evaluated topical EGCG for modulating skin scar formation (Hirci et al., 2025). Another randomized controlled trial (RCT) (n = 50 healthy adults) assessed oral green tea catechins (1080 mg/day GTC with 50 mg vitamin C) over 3 months for UVR-induced skin inflammation (Taylor et al., 2005), and a case study reported positive outcomes using drainage/application of GTE for various complicated infectious, inflammatory, tropical, and postoperative wounds in humans (Prasanth et al., 2019). For striae distensae, a preexperimental clinical trial with a pretest–posttest design was conducted on 36 women aged 18–35 years (Hsu, 2005), where participants received a 3% GTE cream twice daily for 8 weeks (Kumar et al., 2016). In a randomized, double-blind, controlled trial on skin scar formation, 62 human volunteers were treated with topical EGCG or placebo for 1–6 weeks (Ayuningsih et al., 2024), whereas in UVR-induced inflammation, a randomized, double-blind, placebo-controlled trial enrolled 50 healthy adults (18–65 years), with 25 in the active group and 25 in the placebo group, receiving 1350 mg encapsulated GTE (540 mg GTC) plus 50 mg vitamin C or placebo twice daily for 3 months (Asadi et al., 2013). Clinical outcomes showed that in striae distensae, a significant reduction was observed in Imam Nelva Alviera (INA) scores (p < 0.001), indicating improved appearance, with no reported side effects and universal participant satisfaction (Ayuningsih et al., 2024). In skin scar formation, topical EGCG reduced mast cells (weeks 1–3, p ≤ 0.01), increased M2 macrophages, decreased scar thickness (weeks 1–3, p = 0.001), and improved scar elasticity (week 4, p = 0.01), alongside enhanced hydration (p < 0.01) (Xu et al., 2021). In UVR-induced inflammation, oral GTC (1080 mg/day) with vitamin C for 3 months did not significantly reduce skin erythema, leukocyte infiltration, or eicosanoid response to UVR-induced inflammatory challenge (Sarma et al., 2008), contrasting with earlier promising findings (Singh et al., 2011) (Table 5).
Table 5
Summary of clinical studies on green tea/EGCG in wound healing.
| Source | Study Design | Condition Studied | Sample Size (N) | Form of Green Tea/EGCG & Concentration | Route of Administration | Treatment Duration | Key Clinical Outcomes | Safety/Reported Adverse Effects |
|---|---|---|---|---|---|---|---|---|
| (Ayuningsih et al., 2024) | Pre-experimental, pretest–posttest | Striae distensae (SD) | 36 women | 3% green tea extract cream | Topical | 8 weeks | Significant reduction in INA score (p<0.001), improvement in SD appearance | No adverse effects; all participants satisfied |
| (Lian et al., 2014) | RCT, double-blind, controlled | Cutaneous scar formation | 62 volunteers | Topical EGCG | Topical | 1–6 weeks | Reduced mast cells, increased M2 macrophages, decreased scar thickness (P=0.001), improved elasticity (P=0.01), enhanced hydration (P<0.01) | Not specifically reported in citation |
| (Jagdeo & Brody, 2011) | RCT, double-blind, placebo-controlled | UVR-induced skin inflammation | 50 healthy adults (25 active, 25 placebo) | 1350 mg green tea extract (540 mg GTC) + 50 mg vitamin C | Oral | 3 months | No significant reduction in skin erythema, leukocyte infiltration, or eicosanoid response | Occasional mild nausea reported |
| (Soltani et al., 2014) | Case study | Complicated infected, inflammatory, postoperative wounds | Multiple cases | Green tea extract solution (0.5%–5%) | Topical (drainage, gauze balls, surface application) | Not specified (30 days) | Positive outcomes, stimulation of tissue regeneration, effective in injured tissue recovery | No marked skin toxicity |
| (Ma et al., 2024) | RCT, double-blind, controlled | Type 2 Diabetes Mellitus (T2DM) | 60 T2DM patients | EGCG 300 mg | Oral | Not specified | Increased BMI and DBP in A allele carriers of FTO-rs9939609 | Not specifically reported in citation |
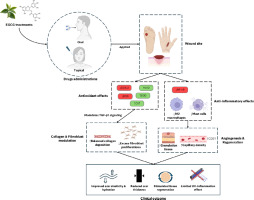
In general wound healing, GTE also proved effective in various complicated infectious, inflammatory, tropical, and postoperative wounds, stimulating tissue regeneration (Bolton, 2016). Regarding safety, oral green tea preparations are generally well tolerated, with mild side effects such as nausea or abdominal discomfort (Desai et al., 2018), although hepatotoxicity concerns arise at high doses (≥800 mg EGCG) or when taken on an empty stomach (Ohishi et al., 2016). The United States Pharmacopeia (USP) recommends avoiding powdered decaffeinated GTE on an empty stomach and discontinuing its use if liver-related symptoms arise, noting that although serious adverse events have been reported, no definitive causal relationship with green tea has been established (Desai et al., 2018). Limited and inconsistent clinical data hinder confirmation of safety and efficacy for wound healing (Tajammal et al., 2025), with methodological flaws such as small samples, short durations, and poor randomization further weakening the evidence (Sen, 2021). Translating findings from animal models to human conditions like diabetic foot ulcers is also challenging (Tajammal et al., 2025), while regulatory approval for chronic wound treatments remains complex and costly (Tajammal et al., 2025). In spite of promising preclinical results, the lack of robust trials prevents clinical adoption, and safety concerns, though generally mild, require careful dosing and monitoring. These gaps represent the most significant barrier to broader commercialization and medical acceptance (Figure 3).
7. MECHANISTIC INSIGHTS
Green tea and its major catechin, EGCG, are potent anti-inflammatory agents that downregulate genes and proteins associated with pro-inflammatory cytokines and enzymes (Hajiaghaalipour et al., 2013). In relation to NF-κB, green tea and EGCG act as antioxidants by scavenging ROS, thereby attenuating NF-κB activity (Ohishi et al., 2016). The Nrf2 pathway is central to this antioxidant action, with EGCG activating HO-1 expression, nuclear Nrf2 translocation, and cytokeratin 16 induction, aiding keratinocyte activation and reepithelialization in diabetic mice (Chen et al., 2017). EGCG also binds KEAP1 to prevent Nrf2 degradation, thereby sustaining Nrf2 signaling and amplifying HO-1 expression, which contributes to enhanced cellular defense and tissue regeneration (Huang et al., 2015). By inhibiting TGF-β1 signaling, EGCG reduces fibroblast proliferation, myofibroblast differentiation, and collagen synthesis, thereby modulating scar formation (Kim et al., 2022). Finally, green tea promotes angiogenesis and granulation tissue formation (Hajiaghaalipour et al., 2013), and modulation of hypoxia-responsive miRNAs contributes to angiogenic regulation (Viaña-Mendieta et al., 2022). EGCG further inhibits the release of TNF-α and IL-6/8/1 through the p38 MAPK and NF-κB signaling pathways (Senger & Cao, 2016).
8. CHALLENGES AND LIMITATIONS
Crude GTEs often yield inconsistent biological activity because of variability in active compound concentrations, seasonal factors, and extraction methods (Tajammal et al., 2025). Many studies rely on partially characterized extracts, limiting reproducibility and obscuring the identification of specific bioactives (Hosseini et al., 2020). Differences in source, processing, and extraction techniques contribute to compositional variability, complicating dose determination, result comparison, and quality control. This lack of standardization poses a major barrier to regulatory approval and clinical adoption. To transition green tea from herbal remedy to pharmaceutical agent, standardized extraction protocols and quantification of key compounds like EGCG are essential. However, green tea polyphenols face challenges of low bioavailability and stability, being sensitive to light, pH, oxygen, and temperature (Al-Rawaf et al., 2019). EGCG’s poor water solubility further limits absorption (Al-Rawaf et al., 2019). Although topical delivery avoids systemic side effects, issues like weak adhesion, limited skin permeability, and potential cytotoxicity at high concentrations require precise control of release rates in wound dressings (Tajammal et al., 2025; Xu et al., 2021).
Although not explicitly detailed in wound healing literature, interindividual variability in response is a well-recognized limitation for natural products (Mostafa et al., 2023). Factors such as gut microbiota composition can influence EGCG metabolism and systemic effects (Capasso et al., 2025). For instance, one study on EGCG in type 2 diabetes found that carriers of a specific FTO gene polymorphism (A allele) demonstrated greater improvements in body mass index (BMI) and diastolic blood pressure (DBP) (Ma et al., 2024), suggesting that genetic variation may modulate therapeutic outcomes. The complex metabolism of polyphenols, affected by host genetics and microbiota, means that standardized doses may not yield consistent therapeutic concentrations or effects across all individuals. This uncertainty complicates clinical trial design and the development of personalized treatment approaches. It underscores the need for pharmacokinetic and pharmacogenomic studies to identify patient subgroups most likely to benefit or requiring dose adjustments, thereby moving beyond a “one-size-fits-all” paradigm.
In spite of promising in vitro and in vivo results, high-quality clinical trial data confirming GTE or EGCG’s efficacy in wound healing remain scarce (Tajammal et al., 2025). Many existing trials face limitations like small sample sizes, short durations, and poor blinding (Hosseini et al., 2020). Replicating complex conditions such as diabetic foot ulcers in animal models is difficult, hindering translation to human use (Tajammal et al., 2025). Regulatory approval for chronic wound dressings is also stringent and costly (Class III devices) (Tajammal et al., 2025). Challenges like extract standardization, low bioavailability, and individual variability further complicate trial design and increase costs, slowing investment and progress. Bridging this translational gap requires more funding and coordinated efforts in formulation science to ensure consistent human efficacy (Table 6).
Table 6
Major challenges and future research directions in green tea/EGCG-based wound healing.
| Category | Major Challenges | Future Research Directions |
|---|---|---|
| Standardization & Composition | Inconsistent biological activity due to variations in active constituents, seasonal effects, and extraction techniques (Tajammal et al., 2025) | Development of rigorous standardization methods for extract composition and specific bioactive compound profiles (Tajammal et al., 2025) |
| Formulation & Delivery | Low bioavailability and stability (degradation by light, pH, O2, temperature); poor water solubility; limited skin permeability; cytotoxicity at high concentrations (Kim et al., 2008) | Nanoformulations (hydrogels, liposomes, nanoparticles, nanofibers) to protect, enhance bioavailability, and enable controlled release (Yu et al., 2024) |
| Response Variability | Inter-individual differences in response (e.g., influence of gut microbiota, genetic variations) (Capasso et al., 2025) | Pharmacokinetic and pharmacogenomic studies to identify patient subgroups and optimize personalized dosing |
| Clinical Evidence | Lack of high-quality human clinical trials, small sample sizes, short durations, methodological limitations (e.g., blinding, randomization), and difficulty replicating complex wound models (Tajammal et al., 2025) | Urgent need for large, well-designed multicenter randomized controlled trials, particularly for chronic wounds such as diabetic foot ulcers (Tajammal et al., 2025) |
| Regulation & Commercialization | Lengthy, costly, and stringent regulatory approval processes, especially for Class III medical devices (Tajammal et al., 2025) | Collaboration among researchers, medical device companies, and regulatory authorities to facilitate research and market approval (Tajammal et al., 2025) |
| Mechanism of Action | Underexplored mechanisms underlying certain effects (e.g., anti-photoaging) (Prasanth et al., 2019) | Further investigation of underlying molecular pathways and target interactions to optimize therapeutic design |
| Toxicity | Potential hepatotoxicity at high doses or when consumed on an empty stomach (Ohishi et al., 2016) | Clear determination of safe dosage ranges and strict usage guidelines, particularly for oral supplements (Hosseini et al., 2020) |
9. FUTURE DIRECTIONS
Nanoformulations and advanced drug delivery systems are key to overcoming the bioavailability, stability, and permeability challenges of green tea polyphenols (Yu et al., 2024). These technologies improve skin permeation, enable controlled release, and protect bioactives from degradation (Ning et al., 2024). Biopolymer-based hydrogels (e.g., chitosan, gelatin) offer flexibility, biocompatibility, and moisture retention, making them ideal for wound care (Yu et al., 2024). EGCG-loaded hydrogels show sustained antioxidant, anti-inflammatory, and antibacterial effects, while promoting tissue regeneration and angiogenesis (Hu et al., 2022). Some even feature self-healing and strong adhesion for use on mobile areas (Yu et al., 2024). Lipid-based carriers like liposomes and transferosomes enhance EGCG delivery and stability (Avadhani et al., 2017). Transferosomes with EGCG and hyaluronic acid improve cell viability and skin permeation (Saghazadeh et al., 2018). Nanoparticles and nanofibers allow sustained release and biocompatibility (Xu et al., 2021). EGCG-Cu2+ nanoparticles exhibit antioxidant, anti-inflammatory, antibacterial properties, and promote vascularization (Kar et al., 2019).
EGCG’s therapeutic efficacy is constrained by its inherent instability and poor bioavailability. As a water-soluble polyphenol, it is prone to oxidation and rapid metabolism, limiting its sustained activity and absorption (Tajammal et al., 2025). These challenges have driven the development of advanced delivery systems, with nanoformulations emerging as a key strategy to protect EGCG and enhance its therapeutic potential. Such technologies aim to elevate green tea from a basic extract to a multifunctional therapeutic agent. Although EGCG-loaded wound dressings have shown promising results in vitro and in vivo, commercialization remains limited because of bioavailability and stability issues (Tajammal et al., 2025). Effective clinical application requires delivery systems that shield EGCG from degradation, enable sustained release, and promote cellular uptake—goals best achieved through controlled, nano-enabled delivery platforms.
Hydrogels are widely recognized as optimal wound dressing platforms because of their high water content, which mimics the ECM and supports moist healing and autolysis (Karamchand et al., 2021). Recent advances have redefined their role, integrating botanical compounds like green tea to create “smart” biomaterials with intrinsic therapeutic functions. Green tea-derived carbon dots (GCDs) have been incorporated into hydrogels, imparting antioxidant, antibacterial, and anti-inflammatory properties (Hu et al., 2024). These multifunctional hydrogels actively contribute to healing, as demonstrated by in vitro assays showing ROS scavenging, cell migration promotion, and excellent biocompatibility (Hu et al., 2024). Electrically conductive GCD-infused hydrogels further accelerate healing in vivo by enhancing angiogenesis, epidermal regeneration, and reducing inflammation (Hu et al., 2024).
A particularly compelling example is a self-healing hydrogel dressing derived from green tea, which showed a 2% higher healing rate and enhanced neovascularization in diabetic animal models (Zawani & Fauzi, 2021). The material effectively exerted its anti-oxidative properties via ROS assay and aided in wound healing, facilitating proangiogenic activity (Zawani & Fauzi, 2021). Another study focused on a bioinspired hydrogel nanocomposite system, which demonstrated fast wound closure through synergistic interactions without inherent toxicity (Zawani & Fauzi, 2021). These findings indicate that the future of green tea in wound care lies in the development of engineered biomaterials that integrate multiple functionalities. The botanical active compound, EGCG, becomes one crucial component of a holistic wound care solution, where the material itself contributes to the therapeutic effect through properties like sustained release and biocompatibility (Zhu et al., 2022). This represents a significant advancement from a simple topical cream to an advanced tissue engineering solution.
Beyond hydrogels, nanoformulations like liposomes, lipid nanoparticles, and electrospun nanofibers are being developed to improve EGCG delivery (Zawani & Fauzi, 2021). EGCG-loaded nanofiber membranes made from polycaprolactone and gelatine effectively counter oxidative stress, showing promise for skin tissue engineering (Zawani & Fauzi, 2021). Novel EGCG nanoparticles (EV NPS), combined with bioinert materials like ascorbic acid, gelatine, and chitosan, demonstrated full-thickness wound healing in vivo within 10 days, with enhanced epithelial thickness, neovascularization, and skin appendage formation (Zawani & Fauzi, 2021). Green tea polyphenol nanospheres (TPN) in PVA/alginate hydrogels also showed superior anti-inflammatory effects and accelerated diabetic wound healing via PI3K/AKT pathway activation (Viaña-Mendieta et al., 2022). The superior in vivo results of these engineered nanoparticles and hydrogels reveal that the pathway to clinical relevance is not merely about proving efficacy in a petri dish but about solving the pharmacokinetic and pharmacodynamic challenges of the compound within a living system (Ramalingam et al., 2022). Nanotechnology provides a critical bridge, protecting the active compound and ensuring its effective and sustained delivery to the target site. This makes it a foundational enabler for future translational research (Table 7).
Table 7
Key nanoformulations and their preclinical outcomes.
| Formulation Type | Active Compounds | Key Mechanism | In Vivo Model | Reported Outcome |
|---|---|---|---|---|
| Hydrogel | Green Tea Carbon Dots (GCDs) | Antioxidant, Anti-inflammatory, Antibacterial, Electrical Conductivity | N/A | Excellent biocompatibility, scavenging of ROS, enhanced cell migration and neovascularization (Hu et al., 2024) |
| Hydrogel | Epigallocatechin-3-gallate (EGCG) | Sustained Release | Rat model | Effective wound healing and skin regeneration; sustained release for >48 hours (Zhu et al., 2022) |
| Hydrogel | Green Tea/EGCG | Self-healing, Proangiogenic | Diabetic in vivo model | 2% higher healing rate, enhanced neovascularization (Zawani & Fauzi, 2021) |
| Hydrogel | Green Tea Polyphenol Nanospheres (TPN) | Sustained release, PI3K/AKT pathway regulation | Diabetic rat model | Superior efficacy in accelerating diabetic wound healing (Viaña-Mendieta et al., 2022) |
| Nanoparticles | EGCG, ascorbic acid, gelatine, chitosan | Bio-compatibility | In vivo model (unspecified) | Complete healing of full-thickness skin wound |
| in 10 days; thicker epithelium and neovascular vessels (Zawani & Fauzi, 2021) | ||||
| Nanofiber Membrane | EGCG, polycaprolactone, gelatine | Scavenging of toxic ROS | N/A | Halted oxidative-stress cell damage, showed potential for tissue engineering (Zawani & Fauzi, 2021) |
The complexity of the wound healing process, a multistage cascade involving hemostasis, inflammation, proliferation, and remodeling, presents a formidable challenge for any single therapeutic agent (Zawani & Fauzi, 2021). While green tea catechins, particularly EGCG, possess a range of beneficial properties that can influence these stages, a single-compound approach may not be sufficient to address the myriad factors that can impede healing, especially in chronic wounds (Karamchand et al., 2021). A promising future direction involves the strategic combination of green tea with other complementary bioactives to achieve synergistic or additive effects. This approach is based on the rationale that a multicomponent formulation can target different phases and pathological conditions of the wound, leading to a more comprehensive and robust therapeutic outcome. Green tea’s antioxidant, anti-inflammatory, and antimicrobial properties are well-established and critical for wound healing (Karamchand et al., 2021; Tajammal et al., 2025) Combining EGCG with other natural compounds may enhance these effects or introduce complementary mechanisms. Functional polypharmacology involves co-delivering bioactives that target distinct healing phases, such as pairing anti-inflammatory agents with those promoting angiogenesis or collagen synthesis. Curcumin and epicatechin exhibit synergistic free radical scavenging, while curcumin–ginger combinations enhance collagen production and reduce nonhealing wounds (Slavova-Kazakova et al., 2021). In spinal cord injury models, curcumin–EGCG combinations improved tissue regeneration and immune responses (Ruzicka et al., 2018). Honey, another potent natural agent, offers antimicrobial and regenerative benefits through its low water activity, acidity, and phenolic content, and can restore antibiotic susceptibility in resistant strains (Yupanqui Mieles et al., 2022). Pairing honey with green tea may provide synergistic infection control and tissue repair. Other combinations, such as Aloe vera with vitamin E, show enhanced wound closure because of complementary anti-inflammatory and antioxidant actions (Khan et al., 2025). Advanced dressings incorporating green-synthesized materials and silver nanoparticles further improve antibacterial efficacy (Neves et al., 2010). Overall, multicomponent natural therapies offer a promising strategy to address the multifactorial challenges of wound healing and improve clinical outcomes.
Combination therapies involving green tea or EGCG offer synergistic benefits for wound healing. A green tea–hyaluronic acid gel enhanced fibroblast activity and gingival healing post-extraction, with increased expression of proliferation and angiogenesis genes (Bobadilla et al., 2019). Hyaluronic acid itself supports cell growth and differentiation (Kim et al., 2018). Green tea has also been used alternately with metronidazole for odor control in malignant wounds, potentially lowering drug resistance (Hsu, 2005). EGCG shows synergy with antioxidants like curcumin, vitamin C, quercetin, genistein, and caffeine in cancer models (Saeki et al., 2018), with vitamin C also aiding skin photoprotection (Jagdeo & Brody, 2011). Incorporating EGCG into biomaterials like collagen, chitosan, or PLGA yields multifunctional dressings with anti-inflammatory and antibacterial effects (Xu et al., 2021). EGCG can activate the AKT/eNOS pathway via 67LR receptor, increasing cGMP and promoting vasodilation. Coadministration with PDE-5 inhibitors like vardenafil sustains cGMP and induces apoptosis, suggesting antitumor potential (Jia et al., 2023).
The main barrier to commercial and clinical adoption of green tea–based wound care products is the lack of high-quality human data. While in vitro and in vivo studies strongly support the therapeutic potential of green tea polyphenols, especially EGCG, the absence of rigorous clinical evidence remains the key bottleneck in translation. Animal studies consistently show accelerated wound closure, reduced scarring, enhanced collagen synthesis, and increased angiogenesis (Hajiaghaalipour et al., 2013). Topical and oral applications of green tea have demonstrated superior healing outcomes in preclinical models (Hajiaghaalipour et al., 2013; Neves et al., 2010). A small clinical case series also reported benefits in human subjects (Gulua et al., 2024). However, without robust clinical trials, these findings are insufficient for regulatory approval (Tajammal et al., 2025). This gap reflects not only scientific limitations but also logistical and financial challenges. Bridging the gap requires a strategic shift toward large-scale, multicenter RCTs, the regulatory gold standard (Shore et al., 2024). Although green tea has been clinically evaluated for other health outcomes (Tajammal et al., 2025), its role in wound healing remains underexplored. Regulatory frameworks typically demand at least two well-controlled studies to establish efficacy (Shore et al., 2024). Progress will depend on collaborative research models—public–private partnerships, academic consortiums, and targeted funding—to support trials and meet regulatory standards, ultimately validating green tea’s clinical potential.
In spite of the promising preclinical evidence for green tea in wound healing, the absence of high-quality human clinical trials remains the most significant barrier to clinical translation (Tajammal et al., 2025). Future trials should focus on RCTs with robust, multisite designs and adequately powered sample sizes to generate statistically significant evidence. The use of well-standardized GTEs or purified compounds such as EGCG is essential to ensure consistency and reproducibility of outcomes. Research should prioritize chronic wounds that are particularly challenging to treat, such as diabetic foot ulcers, where the demand for new therapeutic options is high (Tajammal et al., 2025). Comprehensive evaluation of clinical outcomes, including wound closure rates, scar quality, pain reduction, infection control, and adverse effects, should be performed using objective, standardized methodologies. In addition, pharmacokinetic and pharmacodynamic studies should be integrated to elucidate the bioavailability, metabolism, and interactions of EGCG in humans, optimizing dosing regimens and routes of administration.
The path from preclinical promise to commercial availability for green tea–based wound care products is shaped by complex regulatory frameworks in the United States and Europe, which pose unique challenges for botanical therapies. As a natural compound, EGCG and other green tea bioactives face issues of bioavailability, stability, and cytotoxicity, complicating batch-to-batch consistency—a key requirement for regulatory approval (Hu et al., 2024). These challenges, coupled with high development costs and a historically low clinical trial success rate (under 10%), make green tea an unattractive candidate for many pharmaceutical and medical device companies (Tajammal et al., 2025). Products targeting chronic wounds, such as diabetic foot ulcers, often fall under Class III device classification, demanding the highest regulatory standards (Tajammal et al., 2025). To advance commercialization, collaborative efforts among researchers, industry, and government agencies are essential to support clinical trials and streamline approval processes (Tajammal et al., 2025). Innovative formulations, such as nanoformulations, may help address stability and bioavailability concerns, while clear guidelines for product standardization and testing are critical for building trust with regulators and healthcare professionals.
10. CONCLUSION
Green tea demonstrates significant therapeutic potential in wound healing due to its phenolic compounds, particularly EGCG, which exhibit antioxidant, anti-inflammatory, and antimicrobial activities that support key processes involved in tissue repair. These mechanisms contribute to the regulation of oxidative stress, modulation of inflammation, inhibition of bacterial growth, and promotion of tissue regeneration, making green tea a promising candidate for managing complex wounds such as diabetic ulcers. Advances in formulation strategies, including hydrogel and nanoparticle-based delivery systems, have further improved compound stability and bioavailability in preclinical studies. However, clinical translation remains limited by the lack of large-scale, well-designed human trials. Future progress requires multidisciplinary collaboration, development of advanced delivery platforms, and robust clinical investigations to establish safety, efficacy, and regulatory pathways, enabling green tea to emerge as a validated and practical therapeutic option in modern wound care.
ACKNOWLEDGEMENTS
The authors sincerely thank Aretha Medika Utama, Biomolecular and Biomedical Research Center, Bandung, Indonesia, for their generous contribution of materials that were vital to the successful completion of this study.
AUTHOR CONTRIBUTIONS
P.G.P. was in charge of conceptualization, data curation, validation, and supervising. W.W. did conceptualization, data curation, data interpretation, formal analysis, and methodology. F.T. was concerned with validation, formal analysis, and investigation. N.N. was responsible for data collection, writing, supervising, proofreading, and editing. F.Z.M. looked into visualization, review, and editing.