1. Introduction
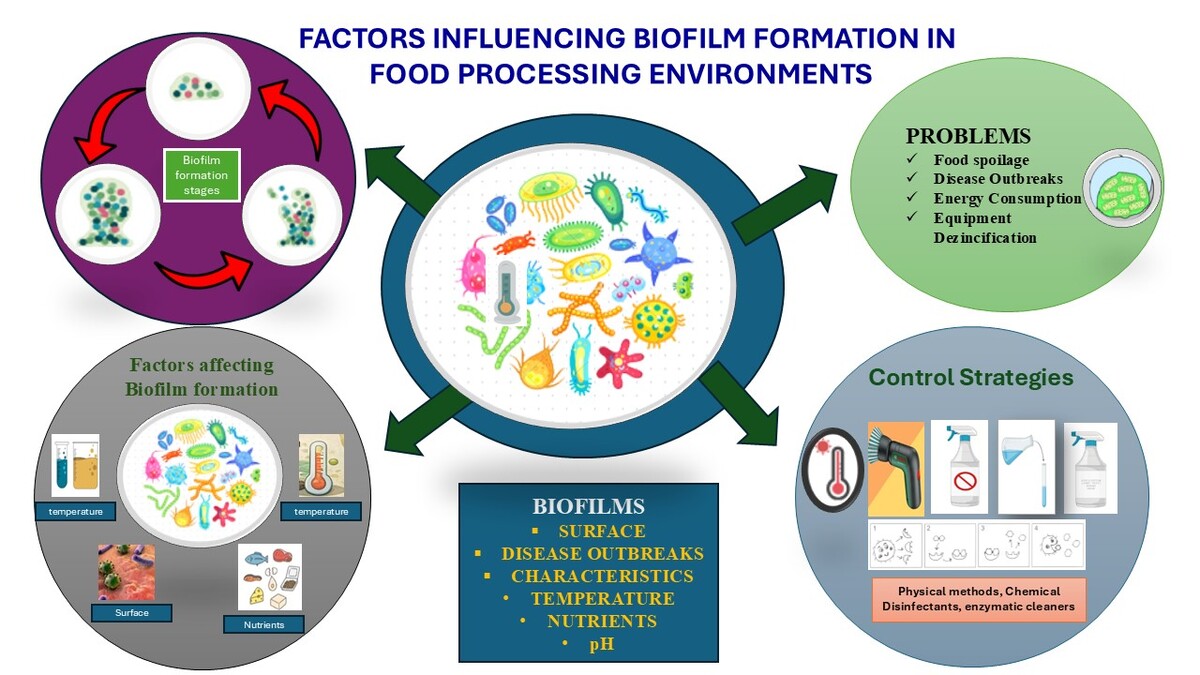
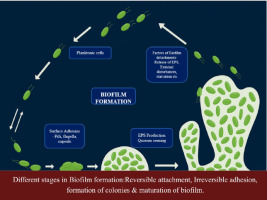
Microbial existence commonly occurs in the form of biofilms, which stand out due to their distinctiveness compared to the planktonic form. They are also much more resistant to environmental factors, antimicrobial agents, and host immune responses (Zanditenas and Ankri, 2024). Understanding the formation, structure, and functions of biofilms is needed to control the impact of this group in different fields, such as healthcare, agriculture, and environmental control (Davey and O’toole, 2000) planktonic (free-swimming; (Velmourougane, Prasanna and Saxena, 2017); They represent the most favorable growth mode for the majority of microbes, especially pathogenic ones (Gottel et al., 2024). Biofilm formation refers to the initial reversible and irreversible microbial attachment to a surface. Biofilm development involves microcolony formation, cellular proliferation, and extracellular polymeric substance (EPS) production, while biofilm maturation denotes the establishment of a structured, three-dimensional community with enhanced resistance and persistence (Hartmann et al., 2021). Although EPS is essential for biofilm development, its production alone does not define biofilm formation, which also requires stable surface attachment and coordinated microbial organization.
Bacterial biofilm development is triggered by the absorption of organic matter (proteins, lipids, polysaccharides, and fatty acids) or inorganic matter (inorganic salts and water). These molecules form an appropriate surface layer, which is then assembled into a diverse arrangement of extracellular polymeric substances (EPS) in single or mixed microbial communities. When bacteria bind to either living or non-living surfaces, they signal each other by means of an extracellular communication system called quorum sensing (QS) (Mukherjee and Bassler, 2019). Biofilm maturation is regulated by QS-mediated intercellular communication. This process involves chemical interactions among bacterial cells and the mechanisms that produce, release, and accumulate autoinducers.
At a certain minimum level, autoinducers trigger signal transduction cascades that cause multicellular responses in microbial populations. This process controls the formation of biofilms, particularly during extracellular polysaccharide production and the formation of channels or columnar structures (Dsouza, Dinesh and Sharma, 2024). These structures ensure the transport of nutrients within a biofilm community. During biofilm development, microorganisms change their phenotypes to adapt to environmental changes or immune responses. Gene regulation is stimulated by regulatory genes and species interactions in multispecies biofilms (Srey, Jahid and Ha, 2013). As a result, QS, EPS, biofilm regulatory genes, and other factors are associated with multispecies biofilm formation. It has been demonstrated that multispecies biofilms are much more resistant to biocides due to interactions between species (Sanchez-Vizuete et al., 2015). In food processing environments, biofilms predominantly develop on food-contact surfaces such as stainless steel pipelines, storage tanks, conveyor belts, gaskets, valves, and heat exchangers, where repeated exposure to moisture, food residues, and incomplete cleaning-in-place (CIP) cycles creates favorable conditions for microbial attachment and persistence. These molecules bind to specific receptors on or within bacterial cells at a concentration indicative of a critical population density.
1.1. Understanding the factors that affect the formation and growth of biofilms in food processing settings is crucial for maintaining food safety and preventing contamination
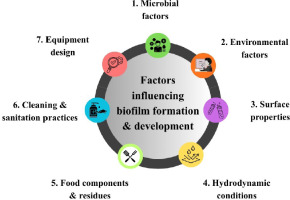
Biofilm formation under food processing conditions is a multifaceted process influenced by a number of factors. These include the nature of surfaces, nutrient availability, temperature, pH level, and the availability of antimicrobial agents. These factors should be systematically recorded along with effective prevention and control mechanisms against biofilm development to ensure food safety and quality in the industry. The process by which biofilms are formed and developed is illustrated in Figure 3, along with the various interrelated factors. All these elements combine to form a complex ecosystem that enables microorganisms to survive and multiply on surfaces. The physical interactions among surface properties, nutrient availability, and communication pathways among microbes are major determinants of the success and fitness of biofilm communities. Environmental factors such as temperature and moisture also regulate biofilm formation and may influence the rate of microbial growth and metabolic activity.
2. Microbial Factors
Within food processing facilities, microbial biofilm formation is strongly influenced by repeated contact with food matrices and by the characteristics of processing equipment, particularly in closed systems such as pipes and tanks, where microorganisms can persist despite routine CIP operations.
2.1. Bacterial species and strains
Some bacterial species and strains vary in their ability to form biofilms, with some exhibiting strong attachment and colonization capabilities. Pseudomonas aeruginosa and Staphylococcus aureus are prominent biofilm formers, particularly in cystic fibrosis lungs and wound infections (Hotterbeekx et al., 2017). Coagulase-negative staphylococci, Enterococcus faecalis, Enterococcus faecium, Escherichia coli, Proteus mirabilis, Klebsiella pneumoniae, and Acinetobacter spp. are other important biofilm formers (Parasion et al., 2014). Polymicrobial biofilms can consist of the fungus Candida albicans and bacteria such as Staphylococcus aureus and Streptococcus mutans (Tabassum et al., 2023). Food safety professionals can develop methods tailored to the biofilm-forming abilities of these microorganisms, which will facilitate the development of novel anti-biofilm technologies, support the maintenance of hygienic environments, and prevent food contamination in the food processing industry.
2.2. Quorum sensing
Much of biofilm formation depends on multiple factors, including quorum sensing (QS), which plays a crucial role in microbial interaction and growth within biofilms. QS is a cell-to-cell signaling pathway that enables bacteria to respond to population density and adjust their behavior (Juszczuk-Kubiak, 2024). QS is associated with disease processes, biofilm formation, and spoilage (Juszczuk-Kubiak, 2024). Pseudomonas aeruginosa is a ubiquitous opportunistic pathogen that utilizes acyl-homoserine lactone (AHL) signals in QS to coordinate gene expression for the production of polysaccharides, rhamnolipids, and other virulence factors (Shrout et al., 2006). Several AHL molecules influence biofilm formation. Biofilm maturation and virulence factor production involve n-butanoyl-homoserine lactone (C4-HSL) and n-hexanoyl-L-homoserine lactone (C6-HSL), whereas biofilm formation is linked to the presence of n-(3-oxododecanoyl)-L-homoserine lactone (3OC12-HSL) (Alayande, Aung and Kim, 2018). However, the relationship between QS and biofilm formation is not always clear. A study of Gram-negative bacteria in a raw vegetable processing line demonstrated no correlation between in vitro biofilm formation and the production of QS molecules (Van Houdt, Hellemans and Volckaert, 2003). Nevertheless, QS has been identified as a major contributor to biofilm formation, particularly under specific environmental conditions. This is demonstrated by the ability of various carbon sources to influence the biofilm patterns of QS mutant strains, such as Las (Lasl/LasR) and Rhl (Rhll/RhlR), which exhibit different biofilm phenotypes depending on the available carbon substrate. In some cases, these mutants show severe defects in initial biofilm formation, whereas in others, they develop biofilms similar to the wild-type strain (Shrout et al., 2006).
2.3. EPS production
EPS production contributes to biofilm development by enhancing adhesion, matrix stability, and protection; however, EPS alone is insufficient to constitute biofilm formation in the absence of sustained surface attachment and microbial organization. Microbial communities are embedded within these matrices, which consist of polysaccharides, proteins, extracellular DNA, and lipids (Flemming et al., 2025). In addition, EPS has applications in technical settings, including wastewater and drinking water treatment plants and water distribution systems, and is associated with biofouling and microbially influenced corrosion (Flemming et al., 2025). EPS aids in the growth of biofilms on food storage tanks and processing lines made of stainless steel during food processing (Drapeau, Marchand and Beaulieu-Prévost, 2012). EPS production enhances cell adhesion, maintains matrix cohesion, and facilitates interactions among cells (Flemming et al., 2025). The preservation is able to reduce shelf life as well as propagate disease (Chmielewski and Frank, 2003). The composition and structure of EPS vary under different environmental conditions. For example, in the presence of heavy metals such as zinc, EPS and soluble microbial product (SMP) polysaccharides are enhanced, indicating increased tolerance to metal toxicity (Redmile-Gordon and Chen, 2017). Such adaptability highlights the complexity of biofilm formation and the challenges associated with its control. Understanding the mechanisms of EPS production is important for developing strategies to regulate biofilms and ensure food safety (Abebe, 2020). In CIP-operated food processing systems, EPS-rich biofilms reduce the penetration of cleaning chemicals and limit shear-induced detachment, making EPS production a critical determinant of biofilm persistence on food-contact surfaces (Chmielewski and Frank, 2003). Further research is needed to optimize enzyme formulations, refine modes of application, and integrate them into existing cleaning systems to maximize their effectiveness in real-world environments. Therefore, EPS should be regarded as a structural and protective component of biofilms rather than as the sole determinant of biofilm formation.
3. Environmental factors
In food processing plants, environmental factors such as temperature, pH, oxygen availability, and nutrient levels vary considerably across processing zones and equipment surfaces, creating localized conditions that directly influence biofilm formation and resistance during production and CIP cycles.
3.1. Temperature
Temperature plays a very important role in biofilm formation during food processing. It has been found to affect the rate and structure of biofilm formation by altering bacterial adhesion. The effect of temperature on biofilm formation is non-linear and strongly dependent on microbial species (Grilo et al., 2021). Biofilm development typically occurs within a defined optimal temperature range, outside of which microbial adhesion, metabolic activity, and extracellular polymeric substance (EPS) production may be reduced or inhibited. An increase in temperature accelerates the rate of biofilm formation in food processing environments. This occurs through several processes. Increased bacterial growth rate: At higher temperatures, the rate at which bacteria reproduce increases, resulting in more rapid colonization of surfaces. Such temperature-dependent biofilm behavior is particularly relevant in food processing equipment, including heat exchangers, cooling tunnels, and storage areas, where temperature gradients may fall within the optimal range for biofilm formation by specific microorganisms.
3.2. Increased metabolic activity
Optimal bacterial motility: Elevated temperatures can enhance bacterial motility, facilitating initial contact with surfaces and subsequent biofilm proliferation. At lower temperatures, motility decreases but still allows biofilm formation (Ma et al., 2019) the major DL concepts pertinent to remote-sensing are introduced, and more than 200 publications in this field, most of which were published during the last two years, are reviewed and analyzed. Initially, a meta-analysis was conducted to analyze the status of remote sensing DL studies in terms of the study targets, DL model (s. STEC strains responsible for producing Shiga toxin formed stronger biofilms observed at 22°C than at 13°C, and a high intensity of biofilm formation occurred at 22 °C within 24 hours, mainly due to several temperature effects:
3.2.1. Cell Surface Properties
Cell Surface Properties: Temperature influences bacterial cell adhesion via its effect on bacterial surface properties, including hydrophobicity. Improved surface characteristics result in increased attachment and biofilm stability at 22 °C compared to 13 °C.
3.2.2. Signal Molecules
Molecules used in quorum sensing, such as c-di-GMP, play a very important role in the formation of biofilms. Their levels increase at 22 °C and promote strong biofilm formation.
3.2.3. Nutrient Hostilities
Intermediate temperatures, such as 22 °C, may boost nutrient absorption and biofilm growth, whereas the reverse may hold at lower temperatures.
3.2.4. Stress Response Mechanism
At 22 °C, reinforcing biofilm structures in response to stress is more efficient, better preparing bacteria to face environmental stress compared to 13 °C. All these factors together lead to stronger biofilm formation of STEC strains at 22 °C, providing them with superior structural and functional integrity compared to lower temperatures (Ma et al., 2019). The optimal temperature differs among bacterial species. Seafood Staphylococcus aureus biofilms are more effectively formed at 37 °C compared to 25 °C, whereas Salmonella sp. forms biofilms at about 30 °C on stainless steel (Speranza, 2019) Temperature is one of the crucial factors in biofilm formation in food manufacturing. Even though growth is faster at higher temperatures, the optimum temperature differs with species. The study of such behaviors is pivotal for the prevention and control of biofilms in food processing plants (Cappitelli, Polo and Villa, 2014; Alvarez-Ordóñez et al., 2019)
3.2.5. pH value
Environmental factors such as pH are vital in biofilm formation and growth during food processing. pH affects bacterial adherence, growth, maturation, structure, and durability of biofilms (Goller, C. C., & RomeoRomeo, 2008). Biofilm development occurs best in food processing environments, where the pH is the most important determinant of biofilm behavior and cleaning resistance (Yuan et al., 2020). The effect of pH depends on the bacterial species and strain; some bacteria create strong biofilms at a specific pH, whereas others are inhibited from doing so (Cappitelli, Polo and Villa, 2014). Such variation emphasizes the multifaceted nature of the biofilm ecosystem and the requirement for specific control measures. Research ought to investigate the interplay of pH with other parameters in order to gain a better understanding of how to regulate biofilm formation in the food industry (Toyofuku et al., 2016). Non-viable bacterial products or metabolic byproducts are known as postbiotics that have been shown to have potential in the control and disruption of biofilms, specifically those formed by pathogens. This is the role of postbiotics in biofilm management.
3.2.6. Biofilm Formation Inhibition
It has been reported that postbiotics from lactic acid bacteria (LAB) could suppress various stages of biofilm formation by influencing surface adhesion, self-aggregation, and quorum sensing of pathogens. These effects disrupt the communication and bonding that support strong biofilm formation, thereby decreasing biofilm integrity (Che et al., 2024a).
3.2.7. Antimicrobial Properties
Postbiotics include compounds such as organic acids, bacteriocins, and other antimicrobial agents that help reduce the growth of biofilms by directly attacking pathogens or by weakening the biofilm structure (Mishra et al., 2024)
3.2.8. Virulence Factor Modulation
Postbiotics (soluble compounds of Lactobacilli) have been demonstrated to suppress the expression of pathogen virulence-associated genes, including those of Aggregatibacter actinomycetemcomitans. This decrease in the capacity of pathogens to proliferate in biofilms disrupts biofilm formation (Ishikawa et al., 2021).
3.2.9. Safety and Stability
Postbiotics are stable and have longer shelf lives compared to live probiotics, and they do not carry the risks that live bacteria can pose, such as infection or the transfer of resistance genes. This allows them to be used in different applications, such as food, biomedicine, and preservation, without safety concerns (Che et al., 2024b; Mishra et al., 2024).
3.2.10. Nutrient availability
Nutrient availability is important in biofilm formation and growth in food processing. It has been demonstrated that the level of nutrients plays a significant role in bacterial communities in biofilms (Xu et al., 2023); (De Plano et al., 2025); (Valiei et al., 2024). High nutrient levels stimulate the production of planktonic cells, whereas low nutrient levels favor biofilm growth (Stanley and Lazazzera, 2004); (Teodósio, Simões and Mergulhão, 2012). The connection between biofilm formation and nutrient levels is complex, as it entails many physiological and environmental factors. As a rule, biofilm formation is a survival mechanism for bacteria and helps bacteria survive under conditions of limited resources, thereby protecting bacteria against environmental factors and antibiotic therapy (Stanley and Lazazzera, 2004).
It can be noted that nutrient levels are suboptimal, and this situation can promote the growth of biofilms more than in nutrient-rich or highly nutrient-poor environments. It has been discovered that moderate to low levels of nutrients can lead to the formation of biofilms by bacteria as a survival mechanism. This is because biofilms have the capacity to act as reservoirs of nutrients and hence trap and make use of resources more efficiently than planktonic cells (Teodósio, Simões and Mergulhão, 2012). Mathematical modeling has indicated that a decreasing concentration of a limiting nutrient in the inflow can decrease the steady-state biomass of planktonic cells. Nonetheless, the proportion of nutrients in the biofilm does not change, and the growth rate of biofilm cells can still be maintained under conditions of a constant exchange rate of the medium, or a constant dilution rate. This may lead to increased accumulation of biofilms over time compared to the planktonic population (Alvarez-Ordóñez et al., 2019).
In addition, bacterial strategies of biofilm production can be influenced by environmental changes, such as variations in nutrient availability. Some bacteria have evolved to be versatile, allowing them to switch between biofilm development and planktonic development, depending on the environmental conditions to which they are subjected. This adaptive mode allows them to take advantage of a changing environment, and transitioning into a growth mode provides them with a competitive advantage over specialized biofilm or planktonic producers (Yan et al., 2017).
The stress of moderate nutrient levels may lead to the acquisition of biofilms as a survival strategy. Adhesion is enhanced in the case of sudden nutrient deprivation, whereas maturation is inhibited in the case of long-term nutrient deprivation (Li et al., 2019). This interwoven connection highlights the importance of maintaining nutrients under strict control to regulate biofilm growth. Knowledge of these interactions assists in developing methods for biofilm prevention and control in the food industry. (Yuan et al., 2020).
3.2.11. Oxygen levels
The effect of oxygen on the development of biofilms is specific to microbial species and the conditions of exposure. Although higher oxygen concentrations can increase biofilm growth in some microorganisms under particular environmental conditions, some species show no effect or even the opposite effect in response to higher oxygen content in their environment. Oxygen concentration is a crucial aspect that contributes significantly to biofilm formation and growth in food processing. Research has shown that aerobic environments can accelerate the rate at which biofilms are formed in certain bacteria. Campylobacter NCTC 11168 biofilms grew more rapidly under aerobic (20% oxygen) compared to microaerobic (5% oxygen, 10% carbon dioxide) conditions, although the level of biofilm formation was the same after three days (Pokhrel et al., 2022; Reuter et al., 2021). The precise impact of oxygen on the formation of biofilms can depend on the bacterial species and the environment in which it resides in the food processing plant. A major study showed that biofilm formation by Campylobacter jejuni, which is a pathogenic bacterium, grows considerably more in an aerobic environment than in a microaerobic environment. This implies that increased oxygen can have a positive effect on the survival and flexibility of some bacteria in response to adverse environmental factors, such as those experienced during food processing (Reuter et al., 2021).
The nature of surfaces on which bacteria attach, such as stainless steel, also plays a role in biofilm formation in food processing environments. Bacteria attach to surfaces and develop biofilms, which is a constant problem for preserving food safety and quality due to environmental conditions (temperature, physical characteristics of the surface, and the availability of oxygen) (Dula, Ajayeoba and Ijabadeniyi, 2021).
Further, it has been observed that oxygen gradients may result in the metabolic differentiation of biofilms, where biofilm cells have diverse metabolic activity. Some bacteria are capable of providing conditions that may allow biofilm growth under low oxygen conditions, which frequently results in antibiotic resistance. For example, under low oxygen conditions, the production of certain metabolites promoting biofilm growth might increase, thereby making the removal of such biofilms in food processing more challenging. Biofilm formation (particularly biofilms resistant to traditional disinfection) may result in severe food safety issues. This is because biofilms not only protect bacteria against antimicrobial agents but can also be a source of persistent contamination if not properly controlled and removed (Yuan et al., 2020). Innovative methods, such as the application of hurdle technology, have been proposed, which involves a combination of physical and chemical treatments as an effective method for preventing and controlling the formation of biofilms in food processing environments (Dawan, Zhang and Ahn, 2025). The specific oxygen requirements of bacterial species in a food processing plant should be considered as part of biofilm control measures. Knowledge of such needs can contribute to the development of more effective cleaning and disinfection guidelines, thereby minimizing the risk of food contamination and maintaining higher food safety.
It is important to address these factors by developing innovative approaches that help prevent food contamination and ensure food safety. This means that Campylobacter jejuni is adapted to biofilm lifestyles in aerobic conditions and thus helps it survive. Oxygen levels alter biofilm formation of bacterial species and strains in varying ways. The availability of oxygen may affect biofilm architecture, composition, and the expression of biofilm formation and virulence factor genes (Toyofuku et al., 2016). Particular attention should be paid to the planning of cleaning and disinfection for food processing equipment and surfaces by addressing the specific oxygen requirements of bacterial species (Alvarez-Ordóñez et al., 2019; Yuan et al., 2020).
3.3. Surface properties
Stainless steel and polymeric materials are the primary food-contact surfaces in processing equipment, and their surface finish, roughness, and chemical properties play a decisive role in microbial attachment, biofilm development, and cleanability under CIP conditions.
3.3.1. Material composition
Biofilm formation is dependent on material composition, especially physical properties (Cappitelli, Polo and Villa, 2014); (Dula, Ajayeoba and Ijabadeniyi, 2021). The properties of the surfaces of various materials prevent or facilitate the formation of biofilms because of their surface properties. For example, surface roughness is vital; rough surfaces may promote microbial attachment under certain conditions, particularly when combined with favorable surface chemistry and low shear stress. It is noted that oral biofilms attach differently to various restorative materials in dental practices than to natural enamel. The physical structure and hardness of these substances have a major impact on the formation and development of biofilms (Gharechahi, Moosavi and Forghani, 2012). Studies on polydimethylsiloxane, a widely used biomedical device material, illustrate that its topographic patterns can minimize bacterial adhesion and biofilm formation. Stainless steel, a common material used in food-processing machinery, is durable and resistant to corrosion. Nonetheless, it has surface characteristics such as energy, hydrophobicity, roughness, and texture that play a major role in bacterial adhesion and biofilm formation. Different researchers have pointed out these factors and the role they play in microbial attachment (Dula, Ajayeoba and Ijabadeniyi, 2021). These conditions influence microbial attachment that is essential for the growth of biofilms (Cappitelli, Polo and Villa, 2014). When using moving-bed biofilm reactors (MBBRs), microbial attachment can be increased by changing the carrier surfaces with polymers, coatings, and chemical groups (Morgan-Sagastume, 2018) Although biofilms are commonly studied to address safety concerns, some studies have identified beneficial roles of biofilms, such as their use in the food industry (Zhao, 2025). For example, microbial biofilms in the food industry have been found to have beneficial effects, including improved fermentation and resistance to adverse conditions (Yao, Mohd Esah and Zhao, 2025). Nevertheless, they present serious problems due to their resistance to washing and disinfection, as they might cause health risks and food contamination (Begić and Josić, 2020; Dawan, Zhang and Ahn, 2025). Carrier materials are very important for the growth and effectiveness of biofilms applied in wastewater treatment.
Polymeric Materials: Polyethylene terephthalate (PET), polyvinyl chloride (PVC), and high-density polyethylene (HDPE) are some of the polymeric materials that have been explored based on their biofilm-supportive properties. The effect of such materials on biofilm formation depends on their surface hydrophilicity that is associated with increased microbial adhesion and biofilm density (Setiyawan et al., 2023).
3.3.2. Design Considerations
Surface properties and design of carriers are important factors that impact carrier performance. EPS can be improved by adding a crimped surface design that is essential for the stable and healthy growth of biofilms (Al-Amshawee, Yunus and Azoddein, 2020). The choice of carrier material plays a significant role in promoting biofilm growth, with material composition playing a crucial role in inhibiting and promoting biofilm growth. Carrier materials are important for the choice and design of biofilm-based wastewater treatment technologies, as they determine biofilm growth and the efficiency of the technology.
3.3.3. Surface roughness
Although increased surface roughness can facilitate microbial attachment by providing protective niches, its effect on biofilm formation is not universal and depends strongly on surface chemistry, material properties, and local flow conditions. It has been shown that rougher untreated stainless steel (SS) surfaces become more prone to biofouling compared to polished, brushed, and electropolished surfaces (Awad, Asker, & Hatton, 2018); (Bezek et al., 2023; Wu et al., 2018). A potential option is the application of food-safe oil-based slippery coatings (FOSCs) that do not eliminate residual oil in surface pores but stabilize it, lowering the apparent roughness and preventing bacterial growth (Awad, Asker, and Hatton 2018). Cold plasma technologies have been explored to produce modified surfaces to render them adhesion-resistant. Although they are not superior to mechanical processes when used alone, mechanical cleaning usually involves methods such as high-pressure spraying and mechanical floor scrubbing. These approaches apply a great deal of mechanical force that destroys and dislodges biofilms. Chemical agents are essential to reduce microbial viability. These agents can increase permeability and break biofilm structures, rendering them more vulnerable to elimination. Chemicals cannot always be used to achieve complete removal, but combinations of chemical cleaning agents have been designed to enhance effectiveness by targeting individual biofilm components (Al-Balushi et al., 2024). Both mechanical and chemical cleaning methods should be used in combination to manage biofilms. This approach ensures that biofilm structures are disrupted and microbial loads on the surface are reduced, thereby improving food safety and contamination prevention in the food processing environment (Dawan, Zhang and Ahn, 2025)
3.3.4. Hydrophobicity
Surface hydrophobicity plays an important role in food processing, biofilm formation, and growth. It has been demonstrated that adhesion and biofilm growth are influenced by the hydrophobic properties of bacterial surfaces and substrates (Di Bonaventura et al., 2008; Teh, Lee and Dykes, 2016; Vázquez-Sánchez, Cabo and Rodríguez-Herrera, 2015). Bacterial adhesion is usually enhanced by highly hydrophobic surfaces that result in greater adhesion. Isolates of Staphylococcus aureus with moderate hydrophobicity are able to develop biofilms on food-processing surfaces (Vázquez-Sánchez, Cabo and Rodríguez-Herrera, 2015). Listeria monocytogenes exhibited better biofilm formation on glass and stainless steel than on polystyrene, and cell surface hydrophobicity was influenced by temperature (Di Bonaventura et al., 2008). Nevertheless, a correlation does not necessarily exist between surface hydrophobicity and biofilm formation. Hydrophilic surfaces could promote more robust biofilms. Investigations of cyanobacterial biofilms showed that biomass development was more influenced by hydrodynamics than surface hydrophobicity (Faria et al., 2021). Cells can be removed more easily from smooth, hydrophilic, and neutral surfaces, and this indicates that these surfaces could be resistant to biofilm formation (Pasmore et al., 2002). These interactions should be understood to implement strategies that prevent and control biofilms in the food processing setting (Cappitelli, Polo and Villa, 2014). Biofilms in food processing environments are usually composed of several bacterial species forming complex communities, including Salmonella enterica and Escherichia coli. Not only can these species create biofilms on biotic and abiotic surfaces, but the interactions may involve physical adherence as well as biochemical and genetic exchanges capable of increasing virulence and resistance phenotypes (Giaouris et al., 2015).
3.4. Hydrodynamic conditions
3.4.1. Flow rate
Flow rate is an important hydraulic factor in biofilm formation in food processing. It has been found that flow rate influences the attachment of microorganisms and growth of biofilms. At low Reynolds numbers, higher flow rates increase biofilm growth by improving the supply of nutrients (Kim, Duizer and Grygorczyk, 2022). The critical shear stress preventing premature biofilm formation is 0.3 Pa on smooth surfaces and 0.9 Pa on rough surfaces. A Reynolds number of 6000 and a shear stress of 0.6 Pa affect biofilm formation in food processing (Teodósio, Simões and Mergulhão, 2012). The structure of biofilms responds to hydrodynamic conditions and nutrients. The availability of nutrients determines biofilm structure up to a certain thickness; thereafter, shear stress resistance becomes a vital factor (Teodósio, Simões and Mergulhão, 2012). Knowledge of the correlation between the hydrodynamic environment and the development of biofilms is crucial for the effective management of biofilms within the food industry.
3.4.2. Shear stress
Hydrodynamic conditions, especially flow rate, are important in food processing and contribute to biofilm formation. Studies indicate that flow rate influences the development of microbial attachment and biofilms. The Reynolds number is the ratio of inertial forces to viscous forces and is an important factor in determining the type of flow that will occur, either laminar or turbulent. The Reynolds number is mathematically expressed as [Re = \frac {\rho v L} {\mu}], where (rho) is the density of the fluid, (v) is the fluid velocity, (L) is a typical linear size (diameter in pipe flow), and (mu) is the dynamic viscosity of the fluid. At low Reynolds numbers, increased flow rates enhance biofilm growth by improving nutrient supply. Conversely, high flow rates can suppress biofilm growth at higher Reynolds numbers, as shear stress increases (Kim, Duizer and Grygorczyk, 2022). Shear stress of 0.3 Pa on smooth surfaces and 0.9 Pa on rough surfaces at threshold levels prevent early biofilm formation. A Reynolds number of about 6000 and a shear stress of 0.6 Pa affect biofilm formation in food processing (Teodósio, Simões and Mergulhão, 2012). Biofilms can adjust to nutrient levels and the hydrodynamic environment. Up to a certain thickness, biofilm architecture is determined by nutrient availability, after which shear stress resistance becomes key (Teodósio, Simões and Mergulhão, 2012).
3.4.3. Stagnant areas
Stagnant hydrodynamic conditions, particularly stagnant regions, play an important role in the formation and growth of biofilms within food processing environments. Flow dynamics have a crucial effect on microbial attachment and biofilm formation (Gomes and Mergulhão, 2021). Under microfluidic conditions, these factors control biofilm growth, whereas biofilms can alter flow velocity by decreasing the flow width (Kim, Duizer and Grygorczyk, 2022). The effect of the hydrodynamic environment on biofilm development depends on environmental conditions and the microorganism. Cyanobacterial biofilm formation in marine environments was enhanced under low shear (4 s−1) compared to higher shear (40 s−1) conditions (Romeu et al., 2019). This implies that areas within the food processing facility that are stagnant may be susceptible to biofilm formation. Stagnant zones, where shear rates are low, are likely to favor biofilm growth and need to be monitored. Platforms should be operated under controlled hydrodynamic conditions to obtain reproducible biofilms that are similar to the target scenarios (Gomes and Mergulhão, 2021).
3.5. Food components and residues
3.5.1. Proteins
Salmonella enterica biofilm: Biovolume of proteins in Salmonella enterica biofilms. The following are the biovolume values of proteins among extracellular biofilm components: proteins had the highest biovolume among the extracellular components in biofilms, with values of 1,295.1 ± 1,294.9 μm3 after 3 hours. The high correlation between the total biofilm biovolume and protein biovolume indicates that proteins are good biofilm markers (González-Machado et al., 2018). The addition of salt under conditions of nutrient limitation favored the formation of biofilms (Li et al., 2019). In conclusion, biofilm growth in food processing depends on proteins, and their presence in residues can lead to growth (González-Machado et al., 2018; Alvarez-Ordóñez et al., 2019)
3.5.2. Carbohydrates
Biofilm formation depends on factors such as food residues, with carbohydrates being the most effective. One of the most important biological factors in biofilm formation and survival is carbohydrates (Alvarez-Ordóñez et al., 2019); (Yuan et al., 2020). They promote the formation of biofilms by providing nutrients and serving as building blocks of the extracellular matrix. A study on biofilms in a meat-processing facility identified carbohydrates in all biofilms analyzed, with 0.5–4.3 μg/cm2 glucose equivalents (Wagner et al., 2021). The availability of nutrients may have a complicated effect on biofilm formation. Nutrient deprivation can promote rapid adhesion, whereas prolonged deficiency may impede development. Nutrient restriction, along with salt-enhanced biofilm formation, demonstrates that environmental stressors interact with nutrients to influence development. Food residues contain carbohydrates that are essential in biofilm formation during food processing (Alvarez-Ordóñez et al., 2019; Yuan et al., 2020; Wagner et al., 2021).
3.5.3. Fats
Food elements and remnants, particularly fats, have a critical influence on the formation and growth of biofilms, as they supply nutrients and favor the growth and adhesion of microorganisms to surfaces (Alvarez-Ordóñez et al., 2019); (Yuan et al., 2020). Fats can induce biofilm growth by providing microorganisms with nutrients (Alvarez-Ordóñez et al., 2019), altering the surface properties, improving bacterial adhesion (Winkelströter et al., 2014; Dula, Ajayeoba and Ijabadeniyi, 2021), and protecting microorganisms against environmental stresses and disinfectants (Yuan et al., 2020); (Carrascosa et al., 2021). Food components have different effects on biofilm formation depending on microbial species and environmental factors. It has been discovered that the addition of salt enhances biofilm production and that nutrient limitation with salt significantly enhances biofilm production in Listeria monocytogenes. Fats are important in shaping and strengthening biofilms in food processing conditions. Biofilms are multifaceted complexes of microorganisms that bind to surfaces and are covered with EPS that protect against environmental stressors, such as sanitizers and antimicrobial agents (Dawan, Zhang and Ahn, 2025). These residues may contribute to the initial adhesion of bacteria to surfaces that serve as a prerequisite for biofilm formation. In the presence of fats, they may modify surface characteristics, including hydrophobicity, which determines cell adhesion. The result may be even greater strength of the biofilm structure, thereby making it difficult to eliminate during cleaning and sanitation (Dula, Ajayeoba and Ijabadeniyi, 2021).
3.6. Cleaning and sanitation practices
3.6.1. Choice of cleaning agents
Biofilm formation and growth depend on many factors, including cleaning agents and sanitation procedures. A variety of cleaning agents with distinct mechanisms are employed to address biofilm accumulation and growth on surfaces. Enzyme Cleaners: Enzymes have the potential to break down the biofilm matrix that is commonly made up of extracellular polymeric substances. In some cases, these agents are used together with surfactants and chelating agents to improve their effectiveness (Anand and Singh, 2013). Enzymatic cleaners have been demonstrated to be effective in destroying biofilms caused by bacteria such as Listeria monocytogenes, especially in combination with disinfectants such as peracetic acid (Fagerlund et al., 2020). In addition to biological and enzymatic approaches, chemically synthesized antibiofilm agents have demonstrated promising activity against microbial biofilms. Cationic lipo-benzamide molecules with twin-nonyl chains have been shown to effectively inhibit biofilm formation and exhibit strong anti-Candida activity by disrupting microbial membranes and biofilm architecture (Jain et al., 2018). Similarly, cationic lipo-oxazole compounds display chain-length-dependent antibiofilm efficacy, highlighting the potential of tailored chemical structures for targeted biofilm control. Such small-molecule antibiofilm agents may complement existing CIP and sanitation strategies by enhancing biofilm disruption on food-contact surfaces (Jain et al., 2017).
3.6.2. Alkaline and Acidic Cleaners
These cleaners can be applied in dairy and food processing facilities because of their capability to decompose organic substances and biofilms. On the other hand, acidic cleaners can dissolve mineral deposits and certain biofilms that oppose the cleaning effect (Fagerlund et al., 2020).
3.6.3. Disinfectants
In post-cleaning, the use of disinfectants such as peracetic acid is important to control the development of biofilms, particularly under conditions prone to Listeria monocytogenes resistance (Fagerlund et al., 2020). Biofilms are controlled by these agents that prevent microbial contamination in food production places (Chmielewski and Frank, 2003; Fagerlund et al., 2020). Conventional agents such as chlorinated alkaline cleaners can be used but do not necessarily remove biofilms. Studies have shown that all-alkaline, acidic, and enzymatic cleaning agents, followed by peracetic acid disinfection, can decrease bacteria in pre-formed biofilms by up to 1.8 log (Fagerlund et al., 2020). The activity of agents depends on biofilm age, concentration, exposure time, and temperature (Fagerlund et al., 2020; Yuan et al., 2020). Cleaning agents may use surfactants to reduce bacterial adhesion by decreasing surface tension, and enzymes combined with surfactants and chelating agents may provide more favorable infiltration into the biofilm matrix (Anand and Singh, 2013) Other strategies are considered innovative approaches to control the growing number of biofilms, such as biocontrol, phage therapy, and cell-to-cell communication disruption (Cappitelli, Polo and Villa, 2014; Kim and Park, no date).
3.6.4. Chelating Agents
These agents attach to metal ions, and this disrupts the structural integrity of biofilms that contain such metal ions. They are frequently used together with other cleaning strategies to increase their efficacy in eliminating biofilms (Kim, Duizer and Grygorczyk, 2022). In food processing settings, periodic cleaning with disinfectants such as peracetic acid is essential to control biofilm formation (Fagerlund et al., 2020). Biofilm management can be further improved by optimizing cleaning procedures in terms of concentrations, exposure times, and temperatures.
3.6.5. Frequency of cleaning
Cleaning and sanitation regimes have a dramatic impact on biofilm development and growth, especially with respect to their frequency (Chmielewski and Frank, 2003; Hebishy et al., 2024). Timing is very important; irregular cleaning permits organic matter and microorganisms to grow, which in turn leads to biofilm growth (Cappitelli, Polo and Villa, 2014). To inhibit biofilm formation, it is possible to increase the frequency of cleaning and minimize the risk of contamination (Chmielewski and Frank, 2003; Yuan et al., 2020). Nevertheless, cleaning can cause microbial resistance when done often unless performed properly. The nature of the environment and the characteristics of materials on which they are located make biofilms even more difficult to clean (Dula, Ajayeoba and Ijabadeniyi, 2021). Regular cleaning is a proactive control measure and is essential to break the life cycle of biofilms at their initial stage of formation. Biofilms may be more difficult to eliminate in the long term when microorganisms are exposed to low doses of non-lethal sanitizers (Alonso et al., 2022). Frequent cleaning is vital for preventing the development of biofilms and maintaining food safety; however, to prevent microbial resistance, it is important to follow proper protocols and rotate sanitizers (Yuan et al., 2020). The use of ozone in cleaning-in-place (CIP) processes could help to improve the cleaning performance in biofilm management (Tirpanci Sivri et al., 2023).
3.6.6. Effectiveness of sanitation procedures
Sanitation and cleaning methods affect the growth of biofilms in food processing. Proper sanitation is essential in controlling the production of biofilms and ensuring food safety (Chmielewski and Frank, 2003; Yuan et al., 2020). Conventional cleaning is usually not adequate to remove biofilms completely from surfaces (Alonso et al., 2022; Yuan et al., 2020). High-pressure sprays and mechanical scrubbers have proven to be highly effective in the removal of biofilms. The success of cleaning biofilms depends on age, surface type, and cleaning agents. Seven-day-old biofilms were more resistant than four-day-old biofilms. Removal was enhanced by higher concentration protocols, repeated treatments, longer exposure duration, and higher temperature, with log reductions of 4.0 to more than 5.5 in seven-day-old L. monocytogenes biofilms (Fagerlund et al., 2020). Better protocols and alternative approaches are promising, such as enzyme-based cleaners, naturally derived antimicrobials, and biofilm prevention by quorum-sensing interference (Jha and Anand, 2023). The combination of different disinfection methods could be more effective at controlling biofilms on surfaces that could be achieved by hurdle technology (Yuan et al., 2020)
3.7. Equipment design
3.7.1. Dead ends and crevices
Food processing equipment design plays a vital role in biofilm formation, particularly dead ends and crevices. These features offer environments in which microorganisms can bind and create biofilms (Alvarez-Ordóñez et al., 2019; Cappitelli, Polo and Villa, 2014). These are isolated sites of microbial colonization in machinery, beyond the reach of cleaning. They accumulate organic matter and moisture and provide a nutrient-rich habitat that facilitates the growth of microorganisms (Hebishy et al., 2024). The surface features of equipment, such as crevices, impact bacterial adhesion and biofilm formation that makes it difficult to eliminate pathogens (Drapeau, Marchand and Beaulieu-Prévost, 2012; Yuan et al., 2020). The combination of influences, including equipment design, can affect biofilm formation more than any single factor (Cappitelli, Polo and Villa, 2014).
3.7.2. Chemical Treatments
It is vital to use sanitizers, disinfectants, acidulants, and enzymes to break the biofilm matrix. Chemicals can prevent bacterial adhesion by disrupting the extracellular polymeric substances that keep biofilms attached to surfaces (Dawan, Zhang and Ahn, 2025).
3.7.3. Surface Modifications
Long-term solutions for the prevention of biofilm development can be achieved by modifying the surfaces of food-contact materials. The adhesion of microorganisms can be reduced by minimizing surface roughness and modifying surface chemistry, thereby minimizing the development of biofilms.
3.7.4. Hurdle Technology
Hurdle technology involves the integration of several interventions effective in controlling biofilms. It is a combination of physical and chemical mechanisms to create an unfavorable environment in which biofilms cannot grow. Knowledge of resistance mechanisms of biofilms to certain disinfectants assists in developing effective solutions in the form of hurdles (Yuan et al., 2020).
3.8. Physical Methods
Thermal treatments, electric fields, or ultrasonic systems are methods that can be used to break biofilm structure and increase the effectiveness of the disinfection process.
3.8.1. Material selection
The choice of materials used in equipment plays a significant role in biofilm formation. Food-contact surfaces are essential when it comes to biofilm development potential (Alvarez-Ordóñez et al., 2019; Cappitelli, Polo and Villa, 2014). Cell attachment and biofilm formation are influenced by surface characteristics, such as chemical and physical properties (Cappitelli, Polo and Villa, 2014). The type of material influences the initial adhesion of bacteria that is the first stage of biofilm formation. Certain materials facilitate microbial attachment, whereas others inhibit it. In biofilm formation on food-contact surfaces, material properties make a major contribution to microbial attachment, and various materials favor or inhibit microbial attachment. Strain-specific adhesion rates are demonstrated on polystyrene surfaces, indicating variation in microbial adhesion. These changes provide obstacles that may suppress bacterial adhesion and consequent biofilm development, thus providing a viable approach to maintain hygienic food-contact surfaces. Material properties in combination with conditions such as temperature and growth media can be more effective than a single factor in the formation of biofilms (Cappitelli, Polo and Villa, 2014).
3.8.2. Accessibility for cleaning
The structure of equipment, and specifically cleaning accessibility, is critical in the process of biofilm formation and growth within food processing environments. The inaccessible parts of poorly designed equipment promote the growth of biofilms that is a challenge to the food industry (Cappitelli, Polo and Villa, 2014; Yuan et al., 2020). The surfaces and topography of equipment have a considerable impact on the formation of biofilms and cleanability (Hebishy et al., 2024). These areas offer bacteria secure environments even when cleaning-in-place (CIP) is used (Hebishy et al., 2024). Biofilms can grow on all materials used in the food industry (Møretrø and Langsrud, 2004). To address this, facility designs should ensure thorough cleaning while minimizing areas that promote biofilms (Chmielewski and Frank, 2003; Winkelströter et al., 2014). Improved equipment design and cleaning procedures can significantly reduce the risk of biofilm contamination in food processing facilities. The food industry must enhance equipment design, implement better cleaning procedures, and consider novel biofilm management techniques, including biocontrol agents, enzymes, and phage, in order to mitigate the risk of biofilms (Cappitelli, Polo and Villa, 2014; Yuan et al., 2020).
3.8.3. Future prospectus and advanced technologies
Microbial characteristics, surface properties, environmental conditions, and food components influence the formation and development of biofilms in such environments (Alvarez-Ordóñez et al., 2019; Winkelströter et al., 2014). Strain properties, compartment medium, signaling molecules, and carrier media are important factors in biofilm formation (Zhao, 2025). Surface properties play an important role in cell adhesion, physiology, and early biofilm formation (Renner and Weibel, 2011). The development of biofilms can be influenced by a combination of factors that can be more significant than each individual factor (Cappitelli, Polo and Villa, 2014), and the development of biofilms can be challenging to predict. For example, Pseudomonas aeruginosa enables foodborne pathogens to persist in polymicrobial biofilms. The factors influencing biofilm formation in food processing are multifactorial and interdependent. These factors need to be understood to develop control strategies. However, the contradictory assertions in the available literature highlight the need for further studies to identify trends and establish predictive models of biofilm growth in food processing settings (Cappitelli, Polo and Villa, 2014).
The existing procedures to eliminate or control undesirable bacterial biofilms in food processing plants include physical methods such as scrubbing and cleaning using high-pressure water jets, ultrasonic treatment, and electrolyzed water; chemical methods including sanitizers and disinfectants (chlorine-based compounds, quaternary ammonium compounds), enzymes to degrade extracellular polymeric substances, and surfactants to destabilize biofilm structure; biosolutions such as bacteriophages that attach to specific bacteria, competitive displacement with useful microorganisms, and quorum-sensing inhibitors to disrupt cell-cell interactions; surface modifications including antimicrobial coatings on food-contact surfaces and nanostructured materials to prevent bacterial adhesion; combination treatments involving the use of multiple methods (e.g., physical cleaning followed by chemical disinfection); prevention plans such as routine cleaning and sanitation schedules, good equipment design to reduce biofilm-prone areas, and hygiene practices with employee training; monitoring and detection through early biofilm detection techniques and periodic environmental sampling and biofilm testing; and new technologies including cold plasma treatments, photocatalytic disinfection, and biofilm-degrading nanoparticles. A combination of such strategies, designed to suit particular food processing conditions and target microorganisms, can be effective in managing and eradicating harmful bacterial biofilms.
Although research is available, there is still a lack of comprehensive studies combining different factors to provide a holistic approach to biofilm dynamics in food processing environments. The factors that affect biofilm formation in such environments include strain features, surface characteristics, environmental factors, and interactions between microbes (Cappitelli, Polo and Villa, 2014). Nevertheless, major gaps remain in the understanding of interactions among these factors and their effects on biofilm development. The literature presents discrepant statements and does not show consistent trends to predict biofilm formation (Cappitelli, Polo and Villa, 2014), which means that interactions among multiple factors may be more important than the effects of individual factors. As an illustration, interactions among microbial species within polymicrobial biofilms, including Pseudomonas aeruginosa and foodborne pathogens, have not been sufficiently studied. The unknown interactions between factors affecting biofilm formation should be explored in future studies, with a focus on examining the ecological complexity of biofilms in food settings (Cappitelli, Polo and Villa, 2014). This approach will play a central role in creating more efficient measures to prevent and control biofilms in food processing facilities, thereby improving food safety and quality (Alvarez-Ordóñez et al., 2019; Winkelströter et al., 2014; Yuan et al., 2020). This gap needs to be addressed to develop effective techniques for preventing and controlling biofilms, thereby enhancing food safety and quality.
The exploration of biofilm-inspired technologies has high potential for applications of biomaterials in environmental remediation. Effective translation of research into practical solutions is possible through the cooperation of microbiologists, materials scientists, and engineers. With further understanding of such systems, new knowledge of the behavior and communication of microbial communities can change the landscape of microbial ecology and the control of infectious diseases.
Conclusion
Important factors include microbial characteristics, surface characteristics to which microorganisms attach, environmental conditions to which they are exposed, and the availability of food materials. Biofilm formation is usually enhanced by these interactions, which, in some cases, outweigh individual effects. Microbial adhesion is greatly influenced by surface properties (hydrophobicity or hydrophilicity). Moreover, the presence of nutrients, which are usually abundant in food processing conditions, provides a suitable substrate on which microbial colonization and the formation of biofilms can occur. Temperature, pH, and water activity are environmental factors that play a central role in biofilm formation and stability. Various microbial species have optimal ranges of such conditions at which individual microbes grow optimally, thereby affecting the biofilm as a whole. Further, relationships between microbes, whether synergistic or antagonistic, contribute to additional structural and functional dynamics of biofilms. Interactions between species may strengthen biofilm structure or render it vulnerable. Biofilms therefore need to be managed effectively through a multifaceted approach that addresses these interactions. Mechanical cleaning has been a standard practice, but traditional disinfection should not be used alone, as biofilms are resistant. Hurdle technology is a layered technique that combines different preservation methods, regulating physical and chemical conditions to prevent the formation of biofilms. Surface modification and the use of natural biochemical agents, including essential oils and enzymes, are promising innovations.