1. INTRODUCTION
Leafy vegetables are among the palatable ingredients of the daily diet in the Indian subcontinent due to their taste and nutritional value. They are the source of nutrientss such as carbohydrates, dietary protein, vitamins, minerals, and dietary fiber, as well as a storehouse of a broad spectrum of bioactive components, mostly secondary metabolites (Kadam et al., 2017). These bioactive components are attributed to their antioxidant, antidiabetic, hypocholesterolemic, hypolipidemic, and antimicrobial properties, which help to reduce the risk of developing chronic diseases and cardiovascular diseases (Genkinger et al., 2004). Chronic disorders like hyperlipidemia, diabetes, and hypercholesterolemia are direct or indirect causes of cardiovascular diseases (Rana et al., 2007).
Basella alba L. (Basellaceae), commonly known as vine spinach, is one of the favorite leafy vegetables in the hot humid tropics of India as well as in tropical Asia and Africa (Tindall, 1983). The variability in morphology and habit in B. alba has recently been addressed through the introduction of three varieties—var. alba, var. rubra, and var. scandens, of which the first two are cultivated varieties and B. alba var. scandens is crop-wild variety, new for science (Das, 2025). The lesser-known, underutilized, more palatable crop-wild variety B. alba var. scandens has been identified from the lower Gangetic plain of West Bengal and is morphologically distinguishable from the other two cultivated varieties. According to ethnopharmacological reports, extracts or formulations of Basella have been used in various countries to treat various ailments as traditional medicine (Kumar et al., 2013).
In this study, attempts have been made to identify the bioactive phytochemicals in the leaves of popular cultivated and crop-wild varieties of B. alba, which confer various health-promoting benefits, using GC-MS analysis.
2. MATERIALS AND METHODS
2.1. Materials
Two varieties of popular leafy vegetable B. alba L. (B. alba L. var. alba and B. alba L. var. scandens Saubhik Das) were taken into consideration, and their taxonomic identity was ratified from Central National Herbarium, Acharya Jagadish Chandra Bose Indian Botanic Garden, Sibpur, Howrah, West Bengal, India. All the chemicals used were of analytical grade.
2.2. Methods
2.2.1. Sample preparation
Fresh leaf samples of each plant specimen were dried in the shade and ground in powder. Leaf powder of 10 g from each sample was extracted with 50 mL of methanol for 48 hours with continuous stirring. The crude organic extracts were partially purified by passing through a Silica gel column with an intercalated layer of activated charcoal. The samples were then concentrated using a rotary evaporator and stored at 4°C.
2.2.2. GC-MS analysis
The GC-MS of methanolic leaf extracts was performed on a Shimadzu (Model GCMS-QP2020) series of GC-MS system (Japan) with auto-sampler. SH-Rtx-5 fused silica capillary column (length 30 m × 0.25 mm ID × 0.25 µm film thickness) was used, and pure helium gas was applied as a carrier gas at a constant flow rate of ±1 mL/min and pressure 100 Kpa. The column temperature was programmed from 50°C to 290°C at 5°C increase/min, then held isothermal for 10 minutes. The injector temperature was set at 250°C, and an autosampler injection volume of 1 μL was injected with splitless mode. The ionization energy was 70 eV with a scan time of 0.3 seconds and a mass range of 45–500 AMU.
The compounds were identified using two methods. Specific bioactive compounds represented by certain peaks in the chromatogram were identified by peak area percentage, based on their retention times comparing with those reported in the literature for SH-Rtx-5 columns. The mass spectrum of GC-MS was interpreted using the National Institute of Standards and Technology (NIST) and Wiley spectral libraries. The chemical identity of the bioactive compounds was confirmed by comparison of the mass spectral fragmentation pattern of an unknown compound with the spectrum of the known component stored in the Willey spectral library and NIST (National Institute of Standards and Technologies, Mass Spectra Libraries) database.
3. RESULTS AND DISCUSSION
3.1. Morphological features
The crop-wild variety (B. alba var. scandens) is morphologically very distinct from the two popular cultivated varieties (B. alba var. alba and B. alba var. rubra). The popular cultivated varieties are non-twining, trailing, or procumbent with thick fleshy stems and large oval leaf, while the crop-wild relative is always twining with a thin cord-like stem and smaller cordate leaf. The crop-wild variety is generally grown in the kitchen garden, consumed locally mainly by growers due to its palatability and nutritive value, but it has yet to achieve wide acceptance.
3.2. GC-MS of methanolic leaf extract
Phytochemical profiling of organic leaf extracts of both cultivated and crop-wild variety (Figure 1) revealed presence of 230 bioactive phytochemicals belonging different chemical classes such as fatty acids and fatty acid derivatives, alkanes, phenolic compounds including flavonoids, different types of terpenes and triterpenoids, phytosterols, steroids, ketones, and organic acid esters (Table 1), of which 72 were found to be present in both the variants. A large number of identified phytochemicals are reported to have antidiabetic or hypoglycemic (Table 2) and hypolipidemic or hypocholesterolemic (Table 3) potentials.
In terpenes and terpenoid content, the crop-wild variety is far enriched (16.04%) than its cultivated relative (6.79%), while it is very much competitive (26.72%) with its cultivated relative (30.75%) in terms of fatty acids and fatty acid derivatives. In terms of phenolics and phytosterols, the cultivated variety (7.93% and 1.19%) showed little superiority over crop-wild variety (3.52% and 1.11%).
Figure 1
GC chromatogram of methanolic leaf extracts of B. alba—(A) cultivated variety (B. alba var. alba) and (B) crop-wild variety (B. alba var. scandens).
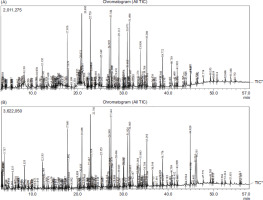
Table 1
Distribution of phytochemical groups in cultivated and crop-wild variety of Basella alba L.
Table 2
List of bioactive phytochemicals with hypoglycemic or antidiabetic effects identified in the organic leaf extracts of cultivated and crop-wild variety of B. alba L.
Table 3
List of bioactive phytochemicals with hypolipidemic effect identified in the organic leaf extract of cultivated B. alba L. and its crop-wild variant.
3.3. Phytochemicals with hypoglycemic hypolipidemic effect
Diabetes mellitus is one of the common health problems in the modern world. Synthetic drugs are commonly applied to combat hyperglycemia, though prolonged use of such drugs may have side effects. Plant-derived bioactive phytochemicals have significant potential for the management of diabetes and hyperglycemia (Balbaa et al., 2021). Ethnobotanical studies have identified nearly 1200 rare plants worldwide that may have antidiabetic potential (Arumugam et al., 2013).
Several terpenoids and diterpenes have been reported as antidiabetic agents, and many have advanced to clinical trials (Mohammed et al., 2022; Panigrahy et al., 2021). Dietary phenolics, especially flavonoids, have demonstrated excellent antihyperglycemic effects (Keshari et al., 2016), inhibiting á-amylase and á-glucosidase enzymes and glucose transporters (Pascual et al., 2015). Terpenoids can inhibit enzymes responsible for the development of insulin resistance and glucose metabolism. Triterpenes appeared promising in the management of diabetes and related problems such as retinopathy, neuropathy, nephropathy, and impaired wound healing (Putta et al., 2016; Mabhida et al., 2018).
Flavonoids have been shown to be therapeutically active in reducing total cholesterol, triglycerides, LDL-cholesterol, and apolipoprotein levels, while increasing HDL-cholesterol, bile acid secretion, and lipid catabolism (Zeka et al., 2017). Terpene derivatives in essential oils have cholesterol-lowering activity (Bahr et al., 2021). Phytosterols may also be very effective in lowering total cholesterol as well as LDL-cholesterol by blocking their intestinal absorption as competitors (Nguyen, 1999).
Dietary supplements are not substitutes for conventional medicine and are not recommended for routine use also, but they can play a vital role in the management of diabetes and hyperlipidemia and their associated problems when used in combination with conventional medicines. According to ethnopharmacological data, more than 800 plant species have been used in the treatment of diabetes in traditional medicine—like bitter melon (Momordica charantia), Fenugreek (Trigonella foenum), Chinese Rhubarb (Rheum palmatum) etc. of which Fenugreek seeds are most promising (Mamgain & Dhrub, 2025). Several plant-derived components have demonstrated significant antidiabetic properties (Sknepnek et al., 2025). A total of 108 human trials on the hypoglycemic effects of herbs and vitamins or mineral supplements have been conducted, which mostly showed greater efficacy when used as adjuncts to conventional treatment (Yeh et al., 2003). Some dietary supplements, such as á-lipoic acid, omega-3 fatty acids, and chromium, are in clinical trials (Martin et al., 2006). The role of dietary supplements such as omega-3 polyunsaturated fatty acids, phytosterols, dietary fibers, and tea catechins in controlling hyperlipidemia has been well demonstrated (Chen et al., 2014). The role of B. alba in the management of hyperglycemia along with several health-related problems, as dietary supplement is substantiated by the presence of a wide array of phytochemicals.
3.4. Basella alba as a source of health-promoting supplements
Vegetables, especially leafy vegetables, are not only a rich source of essential nutrients but also cost-effective, potential herbal remedies with no side effects for managing chronic diseases such as diabetes, hypercholesterolemia, and hyperlipidemia. Proper identification and adequate exploitation of traditional herbs and phytotherapy have been encouraged by World Health Organization (WHO) since 1980.
B. alba is traditionally used to control blood sugar level. The contributory factors for this hypoglycemic effect have yet to be identified. Mucilage present in the plant was considered responsible for that hypoglycemic effect (Palanuvej et al., 2009). Previous studies have also reported its antidiabetic (Nirmala et al., 2009; Bamidele et al., 2014), hypocholesterolemic, and anti-atherosclerotic properties (Bhaskaran et al., 2015). Aqueous extract of B. rubra (400mg/kg body weight for 30 days) was found to significantly reduce the blood sugar level (Nirmala et al., 2011). Appreciable presence of dietary phenolics, terpenes and terpenoids, sterols, fatty acids and fatty acid derivatives, besides mucilage may be contributory factors in lowering blood sugar level as well as the hypolipidemic effect of B. alba. The crop-wild variety appeared more competitive, particularly in terms of phytochemical components.
4. CONCLUSION
The study established the crop-wild variety B. alba var. scandens not only as a palatable substitute for cultivated Basella but also as an herbal medicine. It appeared very competitive, even better than its cultivated popular variety, in terms of phytochemical components—terpenes, terpenoids, and phenolics, including flavonoids, and sterols. Significant presence of such bioactive components may be responsible for its specific medicinal properties in managing hyperglycemia, increased level of blood cholesterol, triglycerides, and LDL-cholesterol.