1. INTRODUCTION
The demand for animal protein, particularly meat and milk products from ruminant production, is projected to rise in accordance with the growing global population over the next 25 years, from the current 6.9 billion to more than 9.7 billion in 2050 (FAO, 2009; Wanapat et al., 2024a). The influence of this message on public discourse regarding hunger and malnutrition has been significant and continues to be so. The FAO’s emphasis on enhancing livestock production significantly contributes to global greenhouse gas (GHG) emissions, primarily methane (CH4). Enteric fermentation, the source of these emissions, accounts for 90% of global GHG emissions from microbial methanogenesis. Ruminal methane production impacts the environment and represents an energy loss for animals, reducing growth rate and milk quality (Wanapat et al., 2024a). For ruminal methanogenesis process, methanogenic archaea including bacteria, protozoa, and anaerobic fungi convert H2 and CO2 through the key enzyme of methyl-coenzyme M reductase (MRC) for CH4 production (Ginovska et al., 2025; Patra & Saxena, 2010). Subsequently, animal sickness caused by pathogen infection such as Mycoplasmopsis bovis-causing severe mastitis disease and cytokine inflammatory activation by interleukin-37 would affect animal productivity and their health (Ellisdon et al., 2017; Gelgie et al., 2024).
Various methane mitigation and risk-reduction strategies for animal sickness have been implemented globally, including the use of feed additives and the application of tropical/marine plant-based phytonutrients (Phupaboon et al., 2025a; Wanapat et al., 2024b,c). Recently, in vitro antimethanogenesis and in vitro antimicrobial trials have shown that the phytogenic compounds (e.g., polyphenols, flavonoids, and antioxidant reagents) derived from plant extracts including garlic oil (Phupaboon et al., 2025b), lemongrass oil (Prachumchai et al., 2024), mangosteen peel (Sommai et al.,
2025), rambutan/red dragon fruit peels (Suriyapha et al., 2025), banana leaf (Dagaew et al., 2024), hemp/marijuana leaf (Phupaboon et al., 2025c), and Mitagyna leaf (Matra et al., 2025). Especially, Moringa oleifera leaf provides several ethnopharmacological applications owing to their potent constituents, including carotenoids, polyphenols, flavonoids, essential amino acids, and phenolic acids, as well as its superior biological efficacy demonstrated in antioxidative reactions through in vitro and in vivo studies (Hashim et al., 2021a,b). Numerous innovative tools and in silico methodologies are being created to enhance the drug discovery process from plant-derived compounds, including molecular docking (e.g., AutoDock Vina, FAFDrugs3, PROTOX, and SwissDock) and pharmacokinetic design (e.g., SwissADME) (Arokiyaraj et al., 2019; Phupaboon et al., 2025a; Stefaniu & Pirvu, 2022). Additionally, the advantages and advancements in science facilitating docking analysis enable scientists to digitally examine chemical databases and select the most potent binders using various scoring algorithms. This study elucidates the binding interaction between two molecules, specifically a ligand (compounds) and a receptor protein of interest, along with friendly safety and pharmacokinetics (Punyauppa-Path et al., 2025).
According to a previous research study, Thai-Moringa leaf extract had the highest potency to decrease in vitro superoxide radical antioxidant capacity and in vivo neuroprotection against oxidative-stress-induced cytotoxicity in SHSY5Y neuroblastoma cells, as well as inhibition of tyrosinase activity (Hashim et al., 2021a,b), while the mode of mechanism was not reported in the effect of ruminal anti-methanogenesis (MRC receptor), anti-mastitis pathogen (Mycoplasmopsis bovis of enolase protein), and anti-inflammatory (interleukin-37 protein). Therefore, this study hypothesized the effects of the selected 40 Moringa-based cytotoxic components on in silico docking analysis with three target proteins.
2. MATERIALS AND METHODS
2.1. Plant collection and extraction
M. oleifera leaves were obtained from a local market in Khon Kaen, Thailand. Clean plant materials were dried in a hot-air oven at 65 °C and subsequently ground. The powdered M. oleifera leaves were subsequently mixed with 70% ethanol at a 1:4 (w/v) ratio and extracted using microwave extraction (Hashim et al., 2021a). The result was a Moringa-based ethanol extract powder for further use.
2.2. Phytochemicals analysis
The phytochemical compositions of Moringa-based ethanol extract were analyzed in terms of antioxidant activities, including total polyphenolic content (TPC) and total flavonoid content (TFC). TPC is reported as mg GAE/g DE, while TFC is expressed as mg QUE/g DE. Following the procedures outlined by Phupaboon et al. (2022), several plant extracts were subjected to the DPPH, ABTS, and FRAP assays, with results presented as mg TROE/g DE. The evaluation of antioxidant activity (expressed as mg TROE/g DE) was performed with three separate methodologies: the DPPH reagent measured at 517 nm, the ABTS reagent measured at 734 nm, and the completed FRAP reagent measured at 593 nm. All analyses were performed in duplicate.
2.3. Biological activity
A part of Moringa-based ethanol extract was tested for its antibacterial efficacy in vitro study because of its powerful antioxidant capability. The minimal inhibitory concentration (MIC) against pathogenic bacteria, including Gram-(-): E. coli TISTR 073, Ent. aerogenes TISTR 1540, S. typhimurium TISTR 292, and Gram-(+): S. aureus TISTR 029, B. cereus TISTR 678, was tested at concentrations of 31.25, 62.5, 125, 250, and 500 µg/mL using a two-fold dilution technique, as per the methodology given by Phupaboon et al. (2025a).
2.4. Moringa-based phytogenic compounds using LC-MS analysis
Based on earlier studies that showed different compounds in Moringa-based ethanol extract, it selected 40 compounds for this study obtained from a liquid chromatograph-quadrupole time-of-flight mass spectrometer (LC–QTOF/MS) analysis, along with their names, molecular formulas, various retention times, mass spectra, and similarity indexes, which are listed in Table 1.
Table 1
Catalog of Moringa-based phytogenic compounds.
2.5. Preparation of ligand molecules and target protein structure
All 40 compounds (Table 1) were chosen as ligand molecules for preliminary screening and to predict the binding site of the energy-minimized target protein structure using the SwissDock server within the SwissDrugDesign platform with AutoDock Vina (https://www.swissdock.ch/). The methods and parameter setup were slightly modified from those described by Phupaboon et al. (2025) and Punyauppa-Path et al. (2025). Ligand molecules were generated via the SwissParam website (http://swissparam.ch/) utilizing the simplified molecular input line entry system (SMILES) derived from the PubChem database.
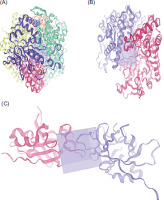
Subsequently, three active site protein targets were selected: methyl-coenzyme M reductase (MRC) of Methanothermobacter marburgensis str. marburg (PDB: 1MRO), Mycoplasmopsis bovis (PDB: 7E2P), and interleukin-37 (PDB: 5HN1). These proteins were retrieved from the RCSB-PDB database, and their three-dimensional (3D) structures and grid box were generated using SWISS-MODEL (Figure 1), according to the procedure of Bugnon et al. (2024).
2.6. Molecular docking prediction
This study assessed the efficacy of 40 selected ligands derived from Moringa-based cytotoxic compounds in inhibiting methanogenesis, combating mastitis pathogens, and reducing inflammation. Each ligand molecule displays unique SMILES structures, as shown in Table 2. This study employed protein active site targets, as illustrated in Figure 1. Molecular docking predictions were performed by evaluating ligand interactions with the active sites of protein target structures using the SwissDock server (https://www.swissdock.ch/) through the AutoDock Vina platform (Bugnon et al., 2024; Punyauppa-Path et al., 2025; Phupaboon et al., 2025a).
2.7. In silico ADME analysis
The in-silico evaluation involved assessing the physicochemical and pharmacokinetic properties of the potent ligands by inputting each SMILES structure into the SwissADME web tool (http://www.swissadme.ch/). This analysis examined physicochemical properties, including lipophilicity (Consensus Log Po/w), water solubility (Log S), pharmacokinetics, and druglikeness in relation to Lipinski’s violations (Phupaboon et al., 2025a).
3. RESULTS
3.1. Phytochemical composition and biological activity
The phytochemical compositions of Moringa-based ethanol extract were 35.2 mg GAE/g DE, 9.2 mg QUE/g DE, as well as 7.0, 31.6, and 62.7 (mg TROX/g DE) in terms of TPC, TFC, DPPH, ABTS, and FRAP, respectively. Importantly, the antimicrobial activity of this extract presented the minimal inhibition concentration (MIC) at 31.25 µg/mL against E. coli, Ent. aerogenes, and S. typhimurium, while the MIC of B. cereus was exhibited at 62.5 µg/mL, as shown in Table 2.
Table 2
Phytochemical composition and antimicrobial activity of Moringa-based ethanol extract
3.2. Molecular docking of ligand binding target protein
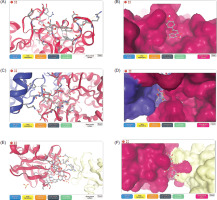
Table 3 illustrates the binding affinity values of the selected 40 ligands binding with three types of target protein, including methyl-coenzyme M reductase (MRC) on subunit alpha protein target of Methanothermobacter marburgensis str. marburg (PDB: 1MRO), Mycoplasmopsis bovis (PDB: 7E2P), and interleukin-37 (PDB: 5HN1) receptors. Interestingly, results showed maximum binding affinity and 3D structure of apigenin ligand with the MRC receptor (PDB: 1MRO) at −7.23 kcal/moL (as shown in Figure 2A,B), along with kaempferol at −7.10 kcal/moL. In terms of tirandamycin-A inhibited the protein of Mycoplasmopsis bovis (PDB: 7E2P) (3D structure in Figure 2C,D), which exhibited the highest affinity value of −8.70 kcal/moL, while cilostazol, lobeline, folic acid, and granisetron were −8.39, −8.23, −8.11, and −8.00 kcal/moL, respectively. In addition, the ligand of 2’’-O-acetylrutin binding with the protein of interleukin-37 (PDB: 5HN1) receptor (3D structure in Figure 2E,F) showed the greater affinity value of −7.94 kcal/moL and −7.18 and −7.05 kcal/moL obtained from folic acid and dicrocin as ligand compounds.
Table 3
Molecular docking of selected LC-MS compounds binding with methyl-coenzyme M reductase (MRC) on subunit alpha protein target of Methanothermobacter marburgensis str. marburg (PDB: 1MRO), Mycoplasmopsis bovis (PDB: 7E2P), and interleukin-37 (PDB: 5HN1).
3.3. Physicochemical and ADME properties of selected ligands
Table 4 presents the selection of three ligands—apigenin, tirandamycin-A, and 2’’-O-acetylrutin—based on their highest binding affinity, for the evaluation of physicochemical and in silico ADME properties in accordance with Lipinski’s rule of five. The physicochemical and ADME results are presented through various parameters, including molecular weight, the count of hydrogen bond acceptors and donors, the number of rotatable bonds, lipophilicity (Log Po/w), water solubility (Log S), pharmacokinetics such as gastrointestinal (GI) absorption, blood–brain barrier (BBB) permeability, P-glycoprotein (P-gp) substrate, and drug-likeness (Lipinski violations and bioavailability score). The results for apigenin and tirandamycin-A ligands were accepted, demonstrating excellent bioavailability in accordance with Lipinski’s rule of five. In contrast, the 2’’-O-acetylrutin ligand was not accepted due to a molecular weight exceeding 500g/moL, as well as having more than 10 hydrogen acceptors and more than 5 hydrogen donors.
Table 4
Physicochemical properties and SwissADME profiles of the selected compounds apigenin and kaempferol.
4. DISCUSSION
As a plant with therapeutic potential and many health benefits, Moringa leaf has been the subject of numerous investigations into its biological activities, including anticancer, antipathogen, anti-inflammatory, and neuroprotective activities. The plant is rich in bioactive phenolic compounds and is widely acknowledged as an essential resource (Hashim et al., 2021a; Shalaby et al., 2022; Said-Al Ahl et al., 2017). The current results confirm the acceptance of apigenin and kaempferol, particularly apigenin, in accordance with Lipinski’s rule. Khusro et al. (2020), who used in silico tools to target MCR receptors to predict the anti-methanogenic attributes of Moringa-based phytocomponents, concur with our findings. Among all the tested phytocomponents, 3,5-bis(1,1-dimethylethyl)-phenol, kaempferol, moringyne, niazimisin, and tetradecanoic acid. The review shows that tetradecanoic acid had the strongest link to the MCR receptor, with a binding energy of −142.98 KJ/moL, followed by niazimicin at −133.98 KJ/moL, kaempferol at −110.36 KJ/moL, 3,5-bis(1,1-dimethylethyl)-phenol at −93.72 KJ/moL, and moringyne at −92.62 KJ/moL. In addition, Dinakarkumar et al. (2021) investigated the application of an in silico molecular docking approach to assess the antimethanogenic properties of phytochemicals derived from Cymbopogon citratus, Origanum vulgare, Lavandula officinalis, Cinnamomum zeylanicum, Piper betle, Cuminum cyminum, Ocimum gratissimum, Salvia sclarea, Allium sativum, Rosmarinus officinalis, and Thymus vulgaris. Under this review, 25 analyzed compounds were assessed for their interaction with MCR protein utilizing the AutoDock 4.0 software; specifically, five compounds are identified: rosmarinic acid (−10.71 kcal/moL), biotin (−9.38 kcal/moL), α-cadinol (−8.16 kcal/moL), and (3R,3aS,6R,6aR)-3-(2H-1,3-benzodioxol-4-yl)-6-(2H-1,3-benzodioxol5-yl)-hexahydrofuro[3,4-c]furan-1-one (−12.21 kcal/moL), and 2,4,7,9-tetramethyl-5-decyn-4,7-diol (−9.02 kcal/moL) exhibited greater binding energy to the MCR protein.
The result of this study indicated that tirandamycin-A, cilostazol, lobeline, folic acid, and granisetron have a greater binding affinity, and tirandamycin-A was accepted in pharmacokinetics and drug-likeness. In accordance with Zahran et al. (2020), it was reported that the selected compounds, specifically tirandamycin derived from marine natural products, exhibited potential inhibitory effects on SARS-CoV-2 targets through molecular docking studies against SARS-CoV-2 RNA-dependent RNA polymerase (RdRp) and methyltransferase (nsp16). Another research study by Adamu et al. (2024) conducted research examining the efficacy of Moringa-based phytochemicals as inhibitors of the Newcastle disease virus (NDV), utilizing molecular docking analysis to assess their anti-pathogenic properties. The review highlights that certain their bioactive compounds strongly attach to a part of the NDV fusion protein, which is crucial for the virus to infect host cells. The review indicated that specific phytochemicals, such as β-sitosterol, catechins, and kaempferol, exhibit significant binding affinity scores of −8.5 kcal/moL, −8.2 kcal/moL, and −7.9 kcal/moL. Additionally, Phupaboon et al. (2025a) demonstrated that the binding affinity of the allicin ligand from microencapsulated garlic oil extract docked with five PDB-protein targets of mastitis pathogens—E. coli, S. aureus, Ent. aerogenes, S. typhimurium, and B. cereus—were –14.99, –14.71, –16.12, –11.14, and -18.89kcal/moL of AC score by SwissDock server through the attracting cavity platform.
Moreover, this research found that the 2’’-O-Acetylrutin ligand strongly reduces the levels of the pro-inflammatory cytokine interleukin-37, along with folic acid and dicrocin. In reviewing the literature, no information was found on how the 2’’-O-Acetylrutin ligand interacts with interleukin-37 in terms of anti-inflammatory activity, using molecular docking and in silico ADME properties. Some publications related to medical plants, including Centipeda minima, Ipomoea pes-caprae, and Dodonaea viscosa, specified phytochemicals alphitolic acid, betulinic acid, malabaric acid, and hispidulin reduced proinflammatory cytokines and cyclooxygenase enzymes (COX-1, 2), with IC50 values ranging from 11.5 to 46.9 µM (Yeshi et al., 2022). Elsewhere, Ali et al. (2023) aimed to identify novel compounds; picrocrocin showed the highest binding affinity of −8.1 kcal/moL when docked against the COX-2 protein, which is associated with various diseases, including inflammation. Another research study by Punyauppa-Path et al. (2025) related to alternative plant-based protein from quinoa natto with Thai herbs used to predict the efficacy of L-leucine ligand docking with different target proteins; type 1 angiotensin II receptor and parathyroid hormone-related protein of Caco-2 cell was −7.65 and −9.15 kcal/moL for AC score.
5. CONCLUSION
To the best of our understanding, Moringa-based polyphenols and/or cytotoxic compounds, which were selected from the 40 components, were potentially used to evaluate in silico, including molecular docking and ADME properties. The main discovery from the molecular docking study showed that the stronger compounds apigenin, tirandamycin-A, and 2’’-O-acetylrutin could help reduce various risks associated with the MRC protein receptor involved in methane production, block the Mycoplasmopsis bovis protein associated with bacteria that cause mastitis, and lower the inflammatory cytokine interleukin-37 linked to mastitis. Additionally, regarding the ADME profile, it was indicated that the potency of the compounds may be acceptable for developing anti-multifunctional drugs for livestock production, particularly in organic meat and milk products in the ruminant field.