INTRODUCTION
Infertility has always been a prime source of stress in society (Ijaz et al., 2022). As per World Health Organization guidelines (Schlatt & Behre, 2010), conventional semen analysis is found to be a crucial diagnostic and therapeutic technique for investigating male infertility. Infertility due to the sperm dysfuction is recognized as changes in sperm motility, morphology and concentration in a sample of sperm analyses. The most conspicuous reason of them is the lower sperm concentration characterized by oligozoospermia, low sperm mobility as asthenozoospermia, and aberrant sperm morphology as teratozoospermia (Kumar & Singh, 2015). Men who have an impairment in sperm parameters collectively known as oligoasthenoteratozoospermia (OAT) syndrome. OAT is mainly due to abnormal spermatogenesis because of environmental, behavioural, dietary and genetic circumstances. Such factors lead to increased chromosomal abnormalities and oxidative stress as a cause of OAT, all of which have a remarkable detrimental effect on the quality of the embryo (Magdi, Darwish, Elbashir, Majzoub, & Agarwal, 2017).
According to WHO statistics, about 60 to 80 million couples worldwide struggling with infertility problems (Ijaz et al., 2022). Large-scale studies have revealed that between 20 and 30 percent cases of infertility are caused by male factors, nearly half of all these are related to female factors, and the remaining 20 and 30 percent are related by causes that are common to both genders (Babakhanzadeh, Nazari, Ghasemifar, & Khodadadian, 2020). Males were more probable to claim infertility assessments when the partners struggle to conceive in Japan than in other countries since couples there tend to oppose adoption or artificial insemination as alternative to bear a child with genes of both parent (Kobayashi, Nagao, & Nakajima, 2012). According to failure of pregnancy during an unprotected sexual intercourse after 12 months, 10 percentage of American partners were deemed to remain infertile (Kumar & Singh, 2015). Infertility affects 13–20% of couples in Poland regardless of race or ethnicity (Walczak-Jedrzejowska, Wolski, & Slowikowska-Hilczer, 2013). Infertility estimates also vary distinctly between Indian states, ranging from 3.7% in Himachal Pradesh, Uttar Pradesh and Maharashtra, 5% in Andhra Pradesh and 15% in Kashmir. According to recent reports based on infertility in India, nearly 50% of infertility cases were caused by male reproductive abnormalities otherwise illnesses (Kumar & Singh, 2015). Based upon the concept that put forward by the Middle East and the North Africans, the female partner is regularly held responsible for infertility. Consequently, men typically decline fertility assessments, which under reports the male infertility (Agarwal, Mulgund, Hamada, & Chyatte, 2015).
The most frequent reason of infertility in males is their inability to generate adequate healthy and active sperms (Dogani, Askari, Kalantari-Hesari, & Rahbar, 2022). There were several prominent explanations for male infertility, crucial one among them are physical causes, hormonal deficiencies, sexually transmitted diseases, environmental, genetic and lifestyle factors (Babakhanzadeh et al., 2020). The notable risk factors allied to male infertility are smoking, age, obesity, alcohol consumption, oxidative stress, various therapeutic drugs (such as antibiotic and antineoplastic agents), reactive oxygen species,varicocele infections, biological or other idiopathic factors, hormonal imbalance that greatly influence the semen quality (Wang, Sudirman, Hsieh, Hu, & Kong, 2020) (Adelakun, Ogunlade, Iteire, & Adedotun, 2021). The normal function of the testicles is hampered by a few external and internal circumstances, including toxins, oxidative stress, diabetes and ageing (Dogani et al., 2022). A rising concern has been expressed in recent years over the harmful outcome of chemicals in developing male reproductive system (Auta & Hassan, 2016). Sperm cells which are highly susceptible to ROS (reactive oxygen species) because of the presence of proportion of poly unsaturated fatty acids in the cytoplasm and plasma membrane, along with their weak antioxidant capacity and DNA repair system. As a result, an alteration in the appearance of sperm, greatly effects the structural and functional integrity (ie, concentration, motility and morphology), suggesting changes in acrosome reactivity and reduction in fertilizing capacity. Moreover, high levels of reactive oxygen species linked to enhanced DNA fragmentation, which is attributed to a low likelihood of spontaneous pregnancies (Ali, Martinez, & Parekh, 2021).
The management upon recognized causes of male infertility were often dealt with a targeted and high success rates. According to idiopathic or genetic bases of male infertility, the medical care is most usually empirical and thus aimed at optimizing fertility (Dabaja & Schlegel, 2014). The utility of empirical medicine for treating idiopathic male infertility is debatable. Important medications like anti-oestrogens, Follicle-stimulating hormone (FSH) and antioxidants therapies are examples of frequently prescribed drugs. It has been suggested that a variety of nutraceuticals, such as fruits, herbs, vegetables, vitamins and nutritional supplements can thereby augment numerous aspects of male fertility. These comprise the parameters for semen analysis and sperm function, erectile function and libido (Ko & Sabanegh, 2014). According to various clinical studies, antioxidant supplements may be utilized to treat male factor infertility caused by oxidative stress. Both patients and clinicians are drawn to these antioxidants because of their low cost and relatively minimal risk of toxicity (Ghanem, Shaeer, & El-Segini, 2010). Particularly , several methods of traditional Chinese medicine (TCM), such as acupuncture and herbal treatments, as well as other traditional approaches, have been used to treat male infertility in animal models or humans. (Khani, Bidgoli, Moattar, & Hassani, 2013). Alternative therapies, like the use of herbal plants, have greater advantages over conventional treatments because they are less invasive and expensive for physical and psychological care. A lot of botanical drugs have a favorable impact on sperm parameters, according to numerous research. People have been familiar with herbal plants since ancient times, however recently, a multifaceted perspective to employ herbal medicines in conjunction with the community care they receive from professionals of health care has established (Mohammadi, Nikzad, Taherian, Mahabadi, & Mahdi, 2013).
In contrast to antioxidants, there are several treatments for managing male infertility in different parts of the world. Recent treatments like ARTs (Assisted reproductive techniques), not too much expensive but it gains a 10–30% success rate (Barati, Nikzad, & Karimian, 2020). In case of In vitro fertilisation, which primarily treats the female spouse, is frequently used as the initial treatment for male infertility (Winters & Walsh, 2014). The unique, even useful method for assessing male fertility is sperm DNA fragmentation. Following In vitro fertilisation and ICSI (Intracytoplasmic Sperm Injection), clinical pregnancy rates are negatively impacted by DNA damage; historically, TESE has only been utilised for males with azoospermia. It has been demonstrated that TESE may be advantageous for sperm with high DNA fragmentation rates (Fainberg & Kashanian, 2019). The most effective treatment for male infertility is ICSI, which enables fertilization with a single spermatozoon that can even be recovered from MESA (Microepididymal Sperm Aspiration) or TESE (Testicular Sperm Extraction) in cases of extreme subfertility. Nevertheless, an excellent likelihood provided by assisted reproductive techniques(ARTs) would not abolish the worth of clinical andrology; rather, it can only change the purpose of doctors, whom we are now expected to boost the fertility potential of a spermatozoal cell thus suitable to improve outcomes in both the IVF (In vitro fertilization ) and the ICSI rather than to treat a likely unidentified reasons of male infertility (Isidori, Latini, & Romanelli, 2005).Unfortunately ICSI and IVF procedures, found to be pricy and upsetting. Furthermore, the utility of ICSI and/or IVF methods can raise the hazard of childhood malignancies and congenital abnormalities in the progeny. As a result, the main option for many infertile couples is still looking for efficient natural therapies to enhance conception (Zhou, Deng, Weng, Weng, & Liu, 2019).
METHODOLOGY
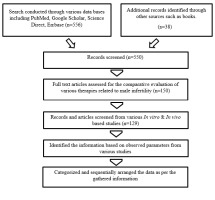
A literature survey was accomplished using, Scopus, Embase, PubMed, Google Scholar, Science Direct to categorize the data’s exploring the role of antioxidants, herbal and synthetic drug therapy (Figure 1). All these shows positive effects on fertility. The search was executed based on subsequent keywords : Infertility, Idiopathic oligoasthenoteratozoospermia (oligozoospermia , asthenozoospermia, teratozoospermia), obesity, oxidative stress, reactive oxygen species, varicocele, hormonal imbalance, diabetes, nutraceuticals, Follicle- stimulating hormone (FSH), anti-estrogens, antioxidants, herbs, vitamins, erectile dysfunction, libido, ART, sperm DNA fragmentation, subfertility, ICSI, TESE, Traditional Chinese medicine (TCM).
RESULTS AND DISCUSSION:
Effect of antioxidants and oxidative stress on the regulation of semen parameters
A disparity among antioxidants and ROS causes oxidative stress, which shows a critical part in the pathophysiology of several manlike diseases (Ali et al., 2021).This imbalance disturbs redox signalling and regulation, which may result in molecular damage. Male infertility cases are on the rise, and oxidative stress is a major contributing factor. Systemic oxidative stress, which is exacerbated by smoking, excessive alcohol use, exposure to various toxins, severe respiratory problems, inflammatory processes or other numerous prolonged disorders, are undeniable factors in the decline of male fecundity (El-Refaei & Abdallah, 2021).
Proper equilibrium of oxidants and antioxidants are indispensable to life. Antioxidants and OS (oxidative stress) can only be understood if the underlying mechanisms that cause oxidation are recognized, together with the activity of both endogenous antioxidants and those made available through dietary intake or supplementation (Cornelli, 2009).
Antioxidants are biomolecules, either naturally occurring or synthesized, that efforts to scavenge free radicals and for promoting their breakdown in the body, or prevent their formation from causing damage. They can give one electron to prevent the undesirable consequences of highly reactive free radicals, or they can change ROS into other less dangerous molecules, to neutralise the effects of the radicals (Ghanem et al., 2010).
Idiopathic male factor infertility and oxidative stress have been allied to several studies. This is primarily because it has been discovered that infertile clients can generate more aberrant spermatozoa, which produce more ROS and less antioxidants, resulting in oxidative stress. Spermatozoa are impacted by oxidative stress in mainly 3 ways, they are : activation of apoptosis, DNA damage and membrane lipid peroxidation. Therefore, many research aim to clarify the efficacy of antioxidant supplementation as a therapy option for infertility (Durairajanayagam, Agarwal, Ong, & Prashast, 2014).
Antioxidant therapy, now suggested as possible approach to reduce the oxidative loss, which manifests as increased sperm DNA fragmentation, decreased mitochondrial function, and compromised acrosome integrity. The antioxidants employed the appropriate concentation of antioxidant, appropriate in vitro incubation time, utilizing co-supplementation with one or more substances are thus found out as the fundamental aspects impacting the improvement of seminal quality by antioxidant supplementation (Adami, Junior, & Losano, 2022).
According to research, incorporating antioxidants such as the vitamins A, B, C, and E in one's dietary regime can boost blood testis barrier stability and can also keep sperm DNA against free radicals (Khaki et al., 2009). The body can be protected against OS by an antioxidant system that includes enzymatic components (Catalase, SOD and glutathione peroxidase),
components which are not enzymatic and compounds with low molecular weight (N-acetylcysteine, glutathione, vitamin E, A, and C, carnitines, coenzyme Q10, lycopene, and others), as well as nutrients (selenium, zinc). Lack of one of them decline the ability of the plasma to fight against free radicals (Walczak-Jedrzejowska et al., 2013).
Antioxidant nutraceuticals in the treatment of iOAT
Nutraceuticals, now extremely popular in treating OAT due to their remarkable availability. Medicative nutraceuticals seems to be widely popular due to growing public knowledge as well as scientific research and consumer interest. Due to their extensive accessibility, nutraceuticals are now quite admired for treating male infertility (Ko & Sabanegh, 2014). Numerous elements of male fertility have been elevated by various nutraceuticals, such as vitamins and nutritional supplements, vegetables and certain herbs (Crimmel, Conner, & Monga, 2001).These elements consist of erectile function, libido, sperm and semen analytical parameters. Numerous flaws exist in the currently available research on nutraceuticals and supplements, including brief length trials, limited sample sizes, inadequacy regarding dose standardization and absence of double blinding or randomization etc (Ko & Sabanegh, 2014) (Table 2; Table 1 ).
Vitamin E (alpha-tocopherol)
Numerous studies have assessed vitamin E as an important chain-breaking antioxidant in membranes (Lombardo et al., 2011). As a result, this antioxidant safeguards the components of the sperm membrane from oxidative damage (Majzoub & Agarwal, 2017).The best vitamin E dose for averting lipid peroxidation in human spermatozoa was determined to be 10 mM (Aitken, Clarkson, & Fishel, 1989). One notable impact based on vitamin E is their potential in lowering ROS levels of sperms in infertile males (Ross et al., 2010). The important source of vitamin E includes cereals, fruits, vegetables, grains, vegetable oils, wheat germ, dairy products, eggs, meat, and poultry (Ko & Sabanegh, 2014). In a study, fifty two patients discovered to take vitamin E and thirty five patients were found to take placebo during the study, that involved 87 people with asthenozoospermia. The study used 100 mg of placebo or vitamin E given in a double-blinded, randomized fashion for 6 months. The findings verified that sperm motility improved with placebo therapy (Suleiman, Ali, Zaiu, El-Malik4, & Nasr, 1996).In a different study, vitamin E's effects in Mancozeb - induced testis injury on mice pups(belongs to first generation) in course of lactation and intrauterine periods were assessed. In this case, vitamin E safeguards the cells from OS by preserving glutathione's natural level as an intracellular free radical scavenger, preventing peroxidation of cell membranes, removing ROS from the body, and depressing apoptosis. Following vitamin E intake, a development in semen parameters were noted, including a rise in sperm quality and quantity as well as a higher sperm survival rate (Saddein, Haghpanah, Nematollahi-Mahani, Seyedi, & Ezzatabadipour, 2019). According to in vitro studies, vitamin E enhances sperm's ability to fertilize in the hamster egg penetration test and prevents sperm from becoming less mobile. In vivo research, individuals with oligoasthenozoospermia, a disorder characterised by a decrease in the amount and motility of sperm cells under the circumstances of OS, responded favorably based on vitamin E treatment (Walczak-Jedrzejowska et al., 2013). GI trouble, fatigue, headache, muscular weakness, visual changes, rash, bruising, and bleeding issues are all possible side effects of Vitamin E (Ko & Sabanegh, 2014).
Vitamin C
It is a water soluble molecule thus shows a tenfold abundance in seminal plasma when comparing with blood serum (Majzoub & Agarwal, 2017). The levels of vitamin C are absolutely related to better fraction of morphologically typical spermatozoa and adversely related to the index of sperm DNA fragmentation (Song, Norkus, & Lewis, 2006).Thus by neutralising, it preserves sperm DNA from oxidative degradation (Greco et al., 2005). Vitamin C can be easily derived from a wide variety of fruits and vegetables, including citrus, papaya, kiwi, strawberries, and so on (Ko & Sabanegh, 2014). In a randomised, double-blinded, clinically controlled experiment, one hundred and fifteen males with clinical varicocele and infertility issues accomplished by defective semen analysis tests were enlisted. All of our findings at these study discovered that vitamin C intake showed a substantial optimistic impact on sperm morphology and motility but not on total count of sperm in young infertile males with poor sperm quality (Cyrus, Kabir, Goodarzi, & Moghimi, 2015). In vitro motility loss at dosage larger than 1000 mmol/L of vitamin C was also verified as a result of interactions with catalytic ions. This further supported the contradictory skill of vitamin C to encourage reactive oxygen species generation (Verma & Kanwar, 1998). As for another investigation with adult male albino rats, 250–300 g body weight with 10–12 months old, on intake of vitamin C reduced infertility due to stress occurred in the uterus by the effects of antioxidants and testosterone rise (Vijayprasad, Ghongane, & Nayak, 2014). In a clinical study with 75 men (20–35 years old) were assigned randomly to 1 of 3 supplementation groups: 200 mg, 1,000 mg or placebo. No improvement in semen quality in the placebo group were noted, while there is greatest development in quality of sperm in the groups receiving ascorbic acid, with 1,000 mg. Heavy smokers who took even more than 200 mg/day of ascorbic acid had better sperm quality (Eb, Wa, We, La, & Mcganity, 1987). Overdose results in headaches, dyspepsia and an increased risk of nephrolithiasis (Ko & Sabanegh, 2014).
Carotenoids
Carotenoids are found to be naturally occuring antioxidant substances which are likely to be present in yellow, orange, red and pink vegetables (Duca, Calogero, Cannarella, & Condorelli, 2019). The combination of carotenoids is usually more effective, despite the fact that the aromatic fat-soluble carotenoid lycopene known as a powerful O2 nullifier .The potent antioxidant in carotenoid family, lycopene, has positive benefits on enhancing male fertility (Babaei, Asadpour, Mansouri, Sabrivand, & Kazemi-Darabadi, 2021). Carotenoids are abundantly present on tomato- containing foods, processed and cooked forms of tomatoes (tomato sauce, ketchup, stewed tomatoes, and so on) that absorb lycopene more efficiently than raw tomatoes (Ko & Sabanegh, 2014). The impact of lycopene on OS caused by varicocele was studied using amimal model. The observations from 45 adult male Wistar rats showed that the varicocele groups had significantly larger range of MDA (Malondialdehyde), ROS, superoxide dismutase (SOD), DNA damage, sperm motility and concentration than the control group (Babaei et al., 2021). Thirty men with idiopathic OAT experienced lycopene treatment. Two milligram of lycopene was administered to patients twice daily for three months. The experts found that 66% patients, had statistically substantial increases in concentration of sperm and also motility (Majzoub & Agarwal, 2018). 30 males with unobstructive iOAT were signed up on a trial and received 2000 mcg of lycopene twice daily for three months. Three months later, the motility, concentration and morphology of sperms were evaluated through analysis of sperm in the semen. Twenty patients (ie, 66%) demonstrated an increase in sperm concentration, 16 (ie, 53%) demonstrated a rise in the mobility of sperms, and 14 (ie, 46%) demonstrated an improvement in morphology of the sperms (Gupta & Kumar, 2002). Excess usage may leads to changes in skin colour, GI distress etc (Ko & Sabanegh, 2014).
Selenium
It has a critical role in spermato-genesis as a trace element. Se is thought to increase the activity of biological glutathione, until now it turns to be a vital element for a set of proteins called selenoenzymes (Majzoub & Agarwal, 2017). Selenium is commonly obtained from various nutritive sources like cereals, nuts (especially Brazil nuts), eggs, seafood and meat products (kidney) (Ko & Sabanegh, 2014).From fifty asthenoteratospermic males with which the semen samples collected and these samples were separated into two groups followed by incubation with 2 g/ml of selenium at 37°C for 2, 4 and 6 hrs. The findings suggested that the in vitro selenium supplementation protects sperm quality amid sampling and preparation of semen, and it is proposed that selenium be added to commercial medium to improve the quality of sperm preparation for ARTs. As a result, the viability and motility were evaluated using 2010 WHO standards (Ghafarizadeh, Vaezi, Shariatzadeh, & Malekirad, 2018). In a double-blind, randomised, placebo-controlled trial was conducted as per cohort study of infertile males with idiopathic oligoasthenoteratospermia, a daily supplementation with 200 g selenium and/or 600 mg NAC (N-Acetyl cysteine) for 26 weeks improved sperm parameters (Safarinejad & Safarinejad, 2009). Potential adverse effects include irritability, GI distress, tiredness and nail changes. Toxic amounts of consumption can cause facial flushing, garlic breath odour, hair loss, metallic taste, tremors, muscle pain, hematological derangements, hepatic and renal failure (Ko & Sabanegh, 2014).
Zinc
The breakdown of RNA and also DNA depends on the essential element zinc. When compared to subfertile men, zinc levels in spermatic fluid were shown to be considerably greater in fertile males (Majzoub & Agarwal, 2017). Zinc is associated with nuclear chromatin condensation, steroidogenesis and testicular growth, acrosin activity, spermatozoa oxygen management, acrosome reaction and finally stabilization of sperm chromatin (Ebisch, Thomas, Peters, Braat, & Steegers-Theunissen, 2007). Zinc insufficiency has been linked to oligospermia, low testosterone levels, and immune system dysfunction (Prasad, 2008). It is clear zinc is present in wheat although it is abundantly present in pumpkin seeds, sunflower, sesame etc (Ko & Sabanegh, 2014). 100 males with asthenozoospermia were randomly assigned to receive either no medication or a zinc treatment of about 250 mg two times a day for about 3 months. In a trial, the patients were monitored for an additional six months, sperm quality significantly improved, as demonstrated by increase in progressive viability and also sperm count (Hassan, 2011; Omu, Dashti, & Al-Othman, 1998). In a set of asthenozoospermic men, oral zinc supplementation successfully enhanced sperm concentration and progressive motility while restoring seminal catalase-like activity (Majzoub & Agarwal, 2018). In another study, the influence of zinc on superoxide anion formation and induction of lipid peroxidation experimentally in infertile men's spermatozoa were investigated in vitro. There was a notable declined activity of zinc on superoxide anion or the O2 generation by human spermatozoa have been observed after ejaculation, hence modifying the properties of sperm cells (Gavella & Lipovac, 1998). Excess of Zn intake might cause loss of appetite, GI issues, stomach ulcers, headaches, dehydration and rash (Ko & Sabanegh, 2014).
Co enzyme Q10
A vital anti-oxidant that is found in nearly all bodily tissues and abundantly present in spermatic mitochondria, thus shows an important role in energy production (Lewin & Lavon, 1997). The levels of CoQ10 were found to be directly related to sperm concentration and motility (Mancini et al., 1994). So CoQ10, although commonly present in soya beans, whole grains, rice bran, almonds, carrots, cabbage, onions, potatoes, oily fish, spinach and organ meat (Ko & Sabanegh, 2014). In a randomized, double-blinded, controlled studies, 228 males suffered with major infertility issues divided equally between the two groups in a random fashion with 1:1 ratio. First group received 200 mg of CoQ10 orally for every day during twenty six weeks, whereas group two received a placebo treatment. The key findings were an enhancement in motility of sperms, density and also their strict morphology (Safarinejad, Safarinejad, Shafiei, & Safarinejad, 2012).
A complete evaluation of 3 clinically controlled randomised trials involving 332 males with infertility issues found that the daily intake of CoQ10 at a dose of 200-300 mg significantly increased sperm concentration and motility. CoQ10, D-aspartic acid zinc incubation considerably reduced sperm LPO in both asthenozoospermic men and normozoospermic patients, and noticeably improved the spermatids motility. As a result, both groups have a considerably greater extent of spermatozoa and thus regained their progressive mobility following swirling of the sperms (Duca et al., 2019). Excess consumption can cause loss of appetite, GI distress, headache and skin rash (Ko & Sabanegh, 2014).
Folic acid
It is a B vitamin (B9) that is essential for synthesizing DNA as well as amino acid breakdown. The ability of the FA (folic acid) in free radical scavenging has led to use as a possible antioxidant for the management of infertility issues in males (Young, Eskenazi, Marchetti, Block, & Wyrobek, 2008). FA is rich in certain dietary sources like brewer's yeast, beans, citrus fruits, enriched cereals, eggs, dark green leafy vegetables, and meat products (Ko & Sabanegh, 2014). In a placebo-controlled, double -blinded, interventional research, 5 mg of folic acid and 66 mg of zinc sulphate daily were administered to 108 mens who had their ability to conceive and 103 subfertile men, thus randomly allocated to undergo 1 of 4 treatments for twenty six weeks. Men with Subfertility showed a massive rise (74%) in total normal spermatozoal count and negligibly 4% expansion in irregular sperms (Wong et al., 2002). In another study, 251 men participating in IVF or ICSI therapy, with subgroups of fertile and subfertile males characterised using spermatozoal count and verified their fertility. Hence, these shows reduced concentration of folate in sperm fluid that was related to higher sperm DNA loss in fertile males (Boxmeer et al., 2009). Excessive consumption can cause GI distress and might cause neurologic symptoms such as irritability, disorientation, sleep disruption and seizures (Ko & Sabanegh, 2014).
Carnitine
A water - soluble antioxidant that is concerned with spermatozoal metabolism and is likely to be an energy source that is actively involved in sperm motility (Majzoub & Agarwal, 2017). Carnitines also deliver energy to gametes and influence spermatozoal maturation and motility (Ko & Sabanegh, 2014). Carnitine is abundantly found in male genitalia, mostly epididymis, and found as marker of epididymal function (Tg, W, & E, 1990). Men suffering from OAT had a considerably diminished levels of carnitine in their sperm, thus L- acetyl carnitine (LAC) and L- carnitine (LC) found to be the most frequently used alternatives in the management of subfertility in males. Carnitine is predominantly present in meats, especially in red meat, fish, chicken and dairy products (Ko & Sabanegh, 2014). Following an in vitro cryopreservation, LC considerably enhanced sperm vitality and motility without disturbing the baseline of sperm DNA oxidations. (Banihani, Agarwal, Sharma, & Bayachou, 2014). In another study that was accomplished on fifty four men suffering with prostate-vesiculo-epididymitis, carnitine 1gm for 3 months improved sperm motility and viability (Vicari, Calogero, & Human Reproduction, 2001). Excessive consumption might cause GI distress, malodorous bodily discharges and convulsions (Ko & Sabanegh, 2014).
Arginine
L-arginine is important for influencing host defenses and cellular immunity. It actively contributes to the production of sperm. L-arginine deficit weakens sperm metabolism, resulting in decreased motility and spermatogenesis (Srivastava, Desai, Coutinho, & Govil, 2006). Since it is vital for sperm capacitation, motility, metabolism and acrosome response (Srivastava & Agarwal, 2010). Many plant-based food contains arginine, containing buckwheat, barley, brown rice, cereals, coconut, chocolate, nuts, dairy and seeds. The daily recommended allowance of arginine is about 20 g, with a maximum dose of about 30 g. Excessive intake may result in GI distress, increased bleeding risk, electrolyte imbalances, renal insufficiency, worsening symptoms of sickle cell disease, elevated glucose levels, asthma, hypotension (Ko & Sabanegh, 2014).
Astaxanthin
A carotenoid derived from Hematococcus pluvialis , an algae (Comhaire et al., 2005). Astaxanthin is found naturally in salmon, algae, yeast, krill, crayfish,shrimp and trout. (Ambati, Moi, Ravi, & Aswathanarayana, 2014) It is also found in various nutritional supplements and thus helps to improve the quality of sperm (Donà et al., 2013). It dramatically reduces ROS formation in seminal fluid and has a faorable impact on certain amount of sperm factors and men's fertility by enhancing linear velocity and also sperm concentration, thus ultimately improving fertilising power. 3-month supplementation with a powerful lipophilic antioxidant, astaxanthin (16 mg/day) or placebo on sperm function and fertility on 30 infertile men were studied utilizing a prospective, double-blind, randomized trial design. The current study reveals that astaxanthin had a good outcome on semen parameters and fertility (Comhaire et al., 2005).
Table 1
Nutraceuticals used in the management of maleinfertility
Nutraceutical anti-oxidant compound | Effect on fertility | Daily dose | Dietary source | Side effects | References |
Vitamin C | Neutralizes free radicals and protects sperm from Oxidative stress | 200-1000 mg | Fruits (citrus, kiwi, papaya, strawberry) and vegetables (broccoli, cauliflower) | Dyspepsia, headache, nephrolithiasis | (Ko & Sabanegh, 2014), (Duca et al., 2019), (Majzoub & Agarwal, 2018), |
Vitamin E | Improves sperm morphology and motility | 100-1200 mg | Fruits, vegetables, cereals, grains, vegetable oils, wheat germ, eggs, dairy, meat, poultry | Fatigue, GI distress, headache, muscle weakness, rash, vision changes, bruising, bleeding complications | (Ko & Sabanegh, 2014) (Majzoub & Agarwal, 2017), (Duca et al., 2019) (Majzoub & Agarwal, 2018) |
Folate/folic acid | Protects sperm DNA production Maintains spermatogenesis. | 400-700 µg | Beans, brewer’s yeast, cereals, citrus, eggs, dark green leafy vegetables, meats | GI distress, rash, irritability, sleep disturbance, confusion, seizures, increased risk of myocardial infarction in those with cardiac history | |
Zinc | Improves testicular development and steroidogenesis spermatozoa oxygen management | 66-500 mg | Seeds (pumpkin, sesame, sunflower ), wheat, meat | GI distress, gastric ulcers, loss of appetite, dehydration, headache and rash | (Ko & Sabanegh, 2014), (Duca et al., 2019), (Ebisch et al., 2007) |
Selenium | Improve sperm count, motility, and morphology Maintain spermatogenesis | 100-225 µg | Cereal, eggs, nuts, meat, seafood | GI distress, fatigue, irritability, nail changes, facial flushing, hair loss, garlic odour on breath, metallic taste, muscle tenderness, tremors, hematologic changes, hepatic and renal insufficiency | (Ko & Sabanegh, 2014), (Duca et al., 2019), (Majzoub & Agarwal, 2017) |
Carnitines | Provide energy for spermatozoa and affect sperm motility and maturation | 3000-4000 mg | Fish, poultry, red meat, dairy products | GI distress, malodorous body secretions, seizures | (Ko & Sabanegh, 2014), (Duca et al., 2019), (Majzoub & Agarwal, 2017) |
Co enzyme Q10 | Protection against oxidative stress induced sperm dysfunction | 200-300 mg | Cabbage, carrots, whole grains, nuts, onions, rice bran, potatoes, soybeans, spinach, oily fish | GI distress, headache, loss of appetite, and skin rash | (Ko & Sabanegh, 2014), (Duca et al., 2019), (Lewin & Lavon, 1997) |
Lycopene | Improves the anti-apoptotic sperm ability Maintain sperm concentration, motility and morphology | 4-30 mg | Tomato | GI distress, changes in skin colour | (Ko & Sabanegh, 2014), (Duca et al., 2019) (Babaei et al., 2021) |
Arginine | Essential amino acid for spermatogenesis Improves sperm motility and concentration. | 20-30 gm | Barley, brown rice, buckwheat, cereals, chocolate, coconut, dairy, nuts, seeds, meats | Asthma, bleeding risk, electrolyte abnormalities, gastrointestinal distress, increased glucose levels, hypotension, renal insufficiency | (Ko & Sabanegh, 2014), (Srivastava et al., 2006) (Srivastava & Agarwal, 2010) |
Astaxanthin | Deteriorates ROS production thus improves sperm quality | 2-4 mg | Shrimp, crab and salmon | - |
Table 2
Effect of antioxidant on specific semen parameters
Herbal therapy in the regulation of male Infertility
Numerous plants, derived as pure molecules and the herbal extracts now established some precautionary effect to a variety of medical conditions, with those that cause damage to the genital system. Contemporary research demonstrated that, administration of the plant extracts has a positive impact on male sperm quality, androgen status, semen parameters and fertility index (Noh et al., 2020). Regarding treatment of male infertility, increased consideration has been given to practice of herbal remedies as complementary medicine or alternative method. Numerous chemical groups, including phytosterols, saponins, carotenoids, oxygenated volatile chemicals, lignans, phenols and alkaloids are among the active phytoconstituents that enhance fertility (Barati et al., 2020). It has been usual all across the world to either improve or decrease male fertility by using a number of medicinal plants with antifertility or fertility enhancing properties. In recent decades, scientific analysis has become concerned in the fertility-related traits of these plants (Roozbeh, Amirian, Abdi, & Haghdoost, 2021).
Infertile couples in most developing nations seek therapy using both natural remedies and modern therapies. Practice of traditional medication often primary treatment option for childless couples in emerging and undeveloped countries due to limited approach to orthodox medications. As a result, even though many plants' or natural products' claims without empirical support, people continue to use herbal treatments, notably among the isolated parts of emerging and underdeveloped communities. Infertility issues are empirically treated with extracts, decoctions, or chemicals that have only partially been purified from medicinal plants. These herbal treatments are employed to treat erectile dysfunction, sexual weakness, loss of libido, ejaculatory dysfunction, and poor sperm production (azoospermia, oligospermia) (Abarikwu, Onuah, & Singh, 2020).
Herbs for managing male infertility
Sesamum indicum
Sesame is recommended by certain traditional doctors to encourage male fertility (Ashamu et al., 2010). In traditional African and Asian medicine, the sesame plant is known for lowering blood cholesterol and treating cancer. Numerous antioxidants, including sesamolin, sesamol, sesaminol diglucoside and sesaminol triglucoside are present in S. indicum. It has a strong likelihood of enhancing fertility (Dada & Adeparusi, 2012). In self-controlled research including 25 infertile participants, examined the influence of sesame on various semen parameters. By consuming sesame for three months dramatically increased sperm count and mobility. Thus sesame's influence on male factor infertility was being studied on people for first time, to our knowledge (Khani et al., 2013). In a separate investigation, thirty adult male Sprague Dawley rats, all in good health, were given leaf extracts of sesame for 6 weeks with doses, 14 mg/kg and 28 mg/kg. However, by a sophisticated hormone interaction close to the male hypothalamic-pituitary-gonadal (HPG) axis and also oestrogen receptor, sesame increases epididymal sperm reserve with enlarged spermatocytes being generated in a dose-related way, as evidenced in this study (Table 3 ).
Withania somnifera
Withania somnifera, in the practice of Ayurveda, also referred to be Ashwagandha ie, poison gooseberry, Indian ginseng or winter cherry. It’s a little perennial shrub with long thick fleshy storage root, is a member of the Solanaceae family (Sengupta et al., 2018). The plant is extensively dispersed in the dry regions of tropical and subtropical zones of Sri Lanka, South Africa, India, Middle East, China and the Canary Islands. In the warmer regions of Europe, it is grown in gardens, and in some regions of Australia, it has turned wild. However, in India W.somnifera cultivated as medicinal herbs. Since ancient times, the whole plant or various sections of it have been utilised for its therapeutic virtues in Indian traditional system of medicines. In Indian Pharmacopoeia, the plant W. somnifera strictly enumerated as official drug (Dar, Hamid, & Ahmad, 2015). The ability of W. somnifera to increase tolerance to stress, decline the ageing process, overall resistance to infections and increase the male reproductive health in conditions like unexplained infertility and psychogenetic impotence has been described in ancient Indian Ayurvedic and Unani medical traditions. W. somnifera is a traditional medication whose roots have been utilized as male sexual stimulant and aphrodisiac in addition to an anti-stress agent (Shukla et al., 2010). 180 male patients undergone infertility issues received W. somnifera powdered root extract with dosage about 5 g each day over the course of three months. This extensive investigation indicated that W. somnifera can possibly treat complex condition of infertility via affecting several enzymatic, metabolic and hormonal processes. However W. somnifera can be recommended to be utilized as an alternate first-hand therapy for the treatment and also preventive medicine for male infertility (Gupta et al., 2013).
Nigella sativa
Nigella sativa, also recognized to be“black seeds”,that belongs to the family Ranunculaceae. Its seed has a black colour and a bitter flavour. The chemical components of N. sativa include thymoquinone (TQ), anthocyanins, flavonoids, alkaloids, linoleic and oleic acid (Mahdavi, Heshmati, & Namazi, 2015). It is grown in the Pakistan, India, Middle Eastern Mediterranean region, South Europe, Syria, Turkey and Saudi Arabia and also indigenous to North Africa, Southern Europe and Southwest Asia. The seeds and oil of N. sativa has extensively utilized over decades to cure a wide range of illnesses all over the world. Additionally, it is a key medication in the indigenous Indian medical systems, including Unani and Ayurveda (Ahmad et al., 2013). Recent research on animals have revealed that N. sativa seed extracts offer an extensive sequence of therapeutic properties, including anti-tumour, anti-microbial, anti-anxiety, anti-inflammatory, anti-oxidant and gastroprotective activity. A beneficial impact was observed on various reproductive indices, undergone in 3 month old male adult Wistar rats (n=24) treated with N. sativa extract of doses 200 and 400 mg/kg at about 60 days (Parandin, Yousofvand, & Ghorbani, 2012).
Punica granatum
The huge deciduous shrub or small tree known as Punica granatum L. (Punicaceae), sometimes known as the pomegranate, utilized therapeutically in the Philippine Islands, Europe, Indo-China, and South Africa. These herbs are traditionally used for various disorders, mainly ulcers, liver damage, snakebites etc (Ajaikumar, Asheef, Babu, & Padikkala, 2005). Pomegranate causes an increase in the amount of epididymal sperm, according to a study done on 30 male Holtzman rats when they were on their child-bearing age about 10 weeks. In order to cure male infertility brought on by lead exposure, P. granatum (pomegranate) may be beneficial. Hence a rise in sperm count implies, strong antioxidant activity of pomegranate, hence found as mediator of the fruit's effect on sperm parameters (Leiva, Rubio, Peralta, & Gonzales, 2011).
Astragalus memberanaceus
Astragalus membranaceus, a commonly used Chinese medicinal plant (Cho & Leung, 2007). The root of A. membranaceus, an annual herb indigenous to the northern domain of China, cultivated largely in China, Indo-China and Korea. A. membranaceus, thus extensively used in East Asia as remedy for different kinds of diseases, and therefore it is expected to possess distinct biological activities, including anti-apoptotic, anti-inflammatory, anti-tumour, anti-oxidant and immune-enhancing properties (Kim, Kim, Chang, & Park, 2016). 5-week-old male mice subjected to root extract of A.membranaceus with doses 100,500,1000mg/kg for 5 weeks shows beneficial effects on testis weight and sperm parameters (Kim, Kim, Park, & Chang, 2012).
Lepedium meyenii
A Peruvian herb called maca (Lepidium meyenii) has been utilised in both human and animal medicine and as a food supplement. Several in vivo investigations have demonstrated that, maca possesses fertility-boosting and spermatogenic properties, sexual behaviour, enhancement of sperm parameters and androgen-like properties (Soo, Won, You, & Ha, 2016). An adult healthy male rats managed orally with an aqueous extract taken from roots of L. meyenii (Maca) for 14 days, give rise to a positive impact on the development of spermatozoa via executing on the first mitosis. Without changing serum levels of testosterone, follicle-stimulating hormone or luteinizing hormone, maca also increased sperm mobility and also total sperm count in healthy men (Gonzales et al., 2004). In a randomized, double-blinded, placebo-controlled parallel investigation lasting 12 weeks, 20 perfectly healthy males were given either maca (1.75 g/day) or placebo treatment. Compared to placebo, sperm number and also sperm mobility showed an increasing trend in maca group. The findings suggested that maca has the ability to increase male fertility (Melnikovova, Fait, Kolarova, Fernandez, & Milella, 2015).
Ruta chalepensis
Ruta chalepensis belongs to Rutaceae family, often called as "ruda" in Mexico, is just a plant that originated from the Mediterranean regions but now it is widely spread all over the world, including Latin America. These herbs are frequently used as treatment options for a variety of complaints including hysteria, vertigo, epilepsy, intestinal worms, colic, poisoning, headache, anxiety and eye issues (Gonzalez-Trujano, Carrera, & Ventura-Martinez, 2006). Although, spermotrophic activity exist in R. chalepensis was altered, and this resulted in a rise in sperm count, sperm motility, decrease in sperm abnormalities, live percentage and a significant increase in FSH (Follicle stimulating hormone) and testosterone with no change in the levels of LH (Luteinizing hormone) and prolactin (Sciences et al., 2022). When mature male rats were given daily doses like 0.5 g, 1.0 g and 2.0 g orally from the aqueous leaf extract of R. chalepensis for 30 days, there was no change in the body weight but a rise in the testicular weights, index, volume and epididymis weight were observed. The plant exhibited a spermotrophic effect, which was shown by a rise in percentage of live sperms, sperm count, sperm motility and a decline in observed spermatozoal defects (Qarawi & A, 2005).
Butea superba
A plant from the Papilionaceae family called Butea superba (Roxb.) indigenous to northern Thailand, has been utilized to improve both physical and mental toughness as well as to prevent age-related health issues. Men's impotence and rejuvenation both have been benefited from its use (Tocharus, Sooksaen, Shimbhu, & Tocharus, 2012). Fifty male Wistar rats , 200–250 g body weight, administered with a daily dose of 2, 25, 250, and 1250 mg/kg body weight of powdered crude drug of B. superba that was suspended in distilled water for 8 weeks, had displayed a tendency of increase in sperm counts and testis weight in rats (Manosroi, Sanphet, Saowakon, Aritajat, & Manosroi, 2006).
Lycium barbarum
The plant genus Lycium barbarum L. is indeed a red coloured fruit belongs to Solanaceae family, utilized as traditionally used Chinese remedy long ago (Li, Ma, & Liu, 2007). Numerous habitual ailments, comprising cancer, hyperlipidemia, diabetes, hypo-immunity function, thrombosis, hepatitis and male infertility, are prevented and treated with L. barbarum fruits due to their wide range of pharmacological activities and functions. L. barbarum fruits showed a substantial advancement in the virtue of spermatozoa, based on several clinical investigations conducted in China (Luo et al., 2006). Young adult healthy male mice were examined for 62 days, after oral administration of saline, sildenafil at a dose of 52 mg/kg (served as standard) and LBP (Lycium barbarum polysaccharide) with doses 10, 20 or 40 mg/kg respectively for analyzing semen parameters. LBP boosted the abundance and complexity of sperms, also significantly raised testicular tissue weights and thus improved the activity of anti-oxidant enzymes. LBP seems to have the ability to increase spermatogenesis in the treatment of major diabetic induced male fecundity problems (Shi et al., 2017).
Morinda officinalis
Morinda officinalis F.C, a well-known herb in China, however recognized an extensive experience of practice, attributed to manage a variety of illness like kidney-yang-deficient disorder, notably male infertility, osteoporosis, polycystic ovary syndrome and depression (Wu et al., 2015). The Northeast Asian traditional medicine has utilised the roots of M. officinalis to treat inflammatory conditions like rheumatoid arthritis and dermatitis as well as impotence and monthly irregularities (Song, Wang, & Wang, 2015). 60 male mice were allocated to three groups randomly and provided with doses of 320, 160, and 80 mg/kg M. officinalis, respectively. An increase in the histological abnormalities and testosterone ranges of testicular tissues were initially reported after treatment with M. officinalis, it somewhat elucidated enhanced mating behavior and sexual urge, and continued the process until 18 days (Wu et al., 2015). Twenty-four Sprague Dawley rats were allocated randomly on 40 g/kg treatment group for 10 days of testing, showed that an aqueous extract from M. officinalis was effective against the damage caused to hypothalamic-pituitary-gonadal axis of male rats by induction of microwave radiation (Song et al., 2015).
Zingiber officinale
Nigeria is among the many Asian and African nations that frequently use the dietary spice ginger (Zingiber officinale) (Morakinyo, Achema, & Adegoke, 2010). There are various biologically active substances found in rhizome of ginger (Z. officinale L.,belongs to Zingiberaceae family), which is frequently used as a condiment which include gingerol, gingerdiol, shogaols, and gingerdione as active constituents. Its anti-tumorigenic, immunomodulatory, anti-apoptotic, anti-inflammatory also antioxidant characteristics are used in various therapeutics. 100 clients for infertility treatment
Participated in a double-blinded, randomized, placebo-controlled experiment. Patients got a 3-month course of oral therapy in which they took ginger and a placebo twice daily in the form of 250 mg capsules. The study showed, ginger successively reduced Sperm DNA Fragmentation in infertile males (Hosseini et al., 2016). In another study, 50 adult male Sprague Dawley rats weighing 200-250 g were administered ginger extract orally in graded doses of 50, 100 and 150 mg/kg body weight for 48 days. According to the observations, ginger administration may even be beneficial for people who have experienced chemotherapy with induced sterility. (Bordbar, Esmaeilpour, Dehghani, & Panjehshahin, 2013).
Solanum nigrum
Africa is extremely popular for native green leafy vegetables like black nightshade (Solanum nigrum L. complex) (Ojiewo et al., 2005). According to reports, the herb contains sedative, diuretic, purgative, antiphlogistic, analgesic, and antiperiodic properties (Zakaria et al., 2006). Twelve to fourteen weeks old, 40 healthy male adult Sprague Dawley rats having a weight about 200–220 g was randomly distributed into four groups (A, B, C and D) of ten rats respectively. For this, the control group ie, group A received daily doses of distilled water equal to 2 ml / kg of body weight and the other groups like B, C and D received a daily consumption of the extract with doses 100, 300 and 500 mg/kg bwt for 28 days accordingly. Rat’s histomorphology, spermatogenesis, steroidogenesis and haematological indices showed that the crude aqueous extract of S. nigrum thus efficiently increases the development and function of the testis (Adelakun, Ogunlade, Olawuyi, Aniah, & Omotoso, 2018).
Phoenix dactylifera
Pollens from date palms (Phoenix dactylifera L. family Palmae) are widely used in the Middle East, particularly in Egypt. In traditional medicine, date palm pollen suspension is frequently used as a folk remedy to treat male infertility (Hassan, 2011). Adult male Wistar rats administered orally with 150 mg/kg dosage of date palm extract aimed at 56 days of treatment showed a positive outcome in the hypothalamic-pituitary gonadal axis which progressively preserved the normal testicular functions and steroidogenesis (El-Kashlan, Nooh, Hassan, & Rizk, 2015).
Satureja khuzestanica
An indigenous plant known as Satureja khuzestanica utilized as an analgesic and antiseptic amongst natives of southern Iran (Tspm, Rastegarpanah, & Ph, 2011) (Amanlou, Dadkhah, Salehnia, & Farsam, 2005). Because of the presence of essential oil in it, it is famous for its medical benefits as antibiotic, analgesic and antiseptic in traditional medicine. Iran's southern region is home to the endemic plant S. Khuzestanica (Tspm et al., 2011). Inhabitants of southern Iran have historically utilised this herb as an analgesic and an antiseptic (Amanlou et al., 2005). Thirty-two male adult Wistar rats were grouped randomly to one of four groups, two of which received Cyclosporine (40 mg/kg/day, orally), and one among these received an oral dose of S. khuzestanica (225 mg/kg/day) about 45 days exposed a substantial development in spermatozoal count, viability accompanied by fertilisation and blastocyst development (Najafi et al., 2016).
Cynamorium coccineum
Cynomorium coccineum L. is a herb belongs to Balanophoraceae family. A parasitic plant with roots that are dark reddish-black and without leaves. It is typically used as a tonic and aphrodisiac and is said to boost semen output (Harraz, 2001). 10 male adult Wistar rats were given 47 mg/100 g/day of aqueous extract of C. coccineum as an oral dosage for 14 days. The outcomes showed that an aqueous extract of the plant significantly increased total sperm number, improved motility, improved fraction of live sperm and lowered aberrant spermatozoa El-Rahman, Haa, Mahmoud, and Harraz (1999).
Ionidium suffruticosum
Ionidium suffruticosum, often known as Hybanthus explants, is an uncommon, sporadic, ethnomedicinal, transient plant, is a member of Violaceae family. Frequently, it is utilized by traditional healers as a tonic and to cure illnesses like diabetes, jaundice, malaria, urinary tract infections, male infertility and water retention (Sonappanavar & Jayaraj, 2011). In a study, sub-fertile male (250 mg/kg body weight) rats were given a 28-day treatment of I. suffruticosum leaf extract, which showed a substantial rise in testicular weight, spermatogenesis, and sperm count (Chenniappan & Murugan, 2017).
Psidium guajava
Psidium guajava is a massive tropical evergreen shrub and a member of the Myrtaceae family. It is indigenous to Mexico and Central America, where it is also widely dispersed. Some researchers focus into the plant's narcotic-like properties, anticough properties, antibiotic, antihyperglycemic, antidiarrheic, antispasmodic, antimutagenic and sedative effects (Uboh, Edet, Eteng, & Eyong, 2010). In a completely randomised design, 30 albino rats that were sexually mature and in excellent health were selected and given oral treatments with mixtures of caffeine and aqueous leaf extracts of guava for 65 days. These considerably boosted total spermatozoal count and viability in the rats (Ekaluo, Ikpeme, Uno, Umeh, & Erem, 2016).
Table 3
Herbal therapy in the regulation of male infertility
Ayurvedic herbal therapy in improving male reproductive health
In Ayurvedic pharmacology, there is a unique class of rasayana herbs known as vajikarana that promote nourishment and also excite the sexual tissues. They also support the promotion of sex appeal and beauty. The term "shukrala" pertains to several categories of spermatogenic- increasing herbs. The right herb can be chosen to satisfy the need if the sources and contributing factors for male infertility are known (Dutta & Sengupta, 2018).
Herbs traditionally used for vajikarana and shukrala purposes
It has been discovered that the sperm supplement kapikacchu (Mucuna Pruriens Bak.), increases sperm mobility and also concentartion. Ashwagandha (Withania somnifera Dunal.), which is thought to exhibit a testosterone-like effect and improves spermatogenesis and Gokshura (Tribulus terrestris Linn.), which increases testosterone levels. Shatavari (Asparagus racemosus Wild.), appears to improve fecundity. Yashtimadhu (Glycyrrhiza glabra Linn.)has been shown to enhance the quality of semen (Table 4 ).
Table 4
Classification of ayurvedic herbal drugs in accordance with their functions on male reproductive health
Synthetic drug therapy in the management of male reproductive health
Medical interventions for recognized causes of male infertility frequently have focused outcomes and high success rates. When male infertility is triggered by idiopathic or hereditary factors, medical intervention tends to become recurrently empirical and aimed at alleviating the situation (Dabaja & Schlegel, 2014). Numerous medications, including androgens, antiestrogens, aromatase inhibitors and rh-FSH, have been attempted in several patients with unknown oligoasthenoteratozoospermia, though it is hardly related to controlled studies, for attaining only a provisional progress in weakened fertility, it may potentially allowing the use of less fiery artificial insemination techniques, such as intrauterine insemination instead of ICSI or IVF, utilizing ejaculated spermatozoa (Table 5).
Aromatase inhibitors
Aromatase inhibitors (AIs) exist as steroidal and non-steroidal oral formulations. In contrast to estrogen receptor modulators, AIs can improve the internal testosterone generation without causing a rise in circulating estrogen levels (Schlegel, 2012). These drugs are used off-label to treat idiopathic OAT (oligoasthenoteratozoospermia) or azoospermia in male patients. Aromatase inhibitors can decrease estrogen levels, raise testosterone levels and thus block the peripheral metabolism of testosterone (Dabaja & Schlegel, 2014). A single instance of induction of spermatogenesis was observed in a man who took a particular dose 2.5 mg/day of letrozole for 4 months and also the testicular biopsy evidence showed that the man's spermatogenesis was induced, and his serum testosterone and FSH levels significantly increased (Schlegel, 2012). All participants in an uncontrolled trial with oral consumption of testolactone and anastrozole (i.e., 74 received testolactone and 104 received anastrozole) reported significant improvement in sperm motility, count and morphology after treatment on infertile oligozoospermic or azoospermic males with aberrant T/E (Testosterone/Estrogen) proportions. (Dabaja & Schlegel, 2014).
Table 5
Synthetic drug therapy for the management of male infertility
Name of the drug | Category | Dose and administration | Duration | Study subjects | Observed effects | Current availibility | References |
Anastrazole | Aromatase inhibitors | 1 mg daily, orally | Mean duration of 4 months | 104 infertile men | Statistically significant increase in T/E ratio and it also improves sperm parameters | Off label use for treating oligospermia and azoospermia | (Dabaja & Schlegel, 2014), (García-Baquero, Fernández-Ávila, & Álvarez-Ossorio, 2020), (Schlegel, 2012), (Chehab, Madala, & Trussell, 2015) |
Letrozole | Aromatase inhibitors | 2.5 mg/day, orally | 6 months | 27 Infertile male patients with low serum testosterone levels and 4 men with non obstructive azoospermia | Increased sperm concentration ,motility and morphologically normal sperm concentration, morphologically normal sperm and ejaculate were observed | used off label for treating oligospermia and azoospermia | (Dabaja & Schlegel, 2014), (García-Baquero et al., 2020), (Schlegel, 2012), (Chehab et al., 2015) |
Testolactone | Aromatase inhibitors | 2 g/day, orally | 8 months | 25 men with assigned randomized controlled cross over trial in patients with idiopathic IOAT | High dose testolactone treatment may actually inhibits T production, so it may not be effective in improving sperm production and fertility | Not approved in USA | |
Tamoxifen | Anti-oestrogen/selective oestrogen receptor modulators | 20 mg/24hr | 3 months | Randomized controlled trials in men with oligospermia/azoospermia, | Improvement in sperm parameters and pregnancy rates | Off label use | (Dabaja & Schlegel, 2014), (García-Baquero et al., 2020), (Nada et al., 2014) |
Clomiphene citrate | SERM | 20 mg/day 25 mg/day, orally | 3-6 months 3 months | 60 patients suffering from idiopathic OAT Total of 197 men undergoes multicentric double blinded randomized trial having infertility and oligospermia | Improved total sperm count,motility and functional sperm fraction Statistically significant increases in sperm concentration and pregnancy rates | Off label use Off label use | (Dabaja & Schlegel, 2014), (Chehab et al., 2015), (Bridges et al., 2015) |
Toremifene/raloxifene GnRH | Non-steroidal oestrogen receptor antagonist Hormonal therapy | 60 mg/day, orally 5-20 μg per pulse through subcutaneous infusion pump | 3 months Every one to two hours | Randomized control trial in patients with oligoasthenozoospermia Infertile men with hypogonadotropic hypogonadism | Improvement in semen parameters and also increased pregnancy rates Improves spermatogenesis | Off label use Only in specialty centers(GnRH was progressively abandoned for its uncomfortable route of administration) | |
rhFSH | Hormonal therapy | 50,100,200IU,SCim | Observed at different time interval(2,3,4 and 5 months) | A total of 354 men affected by idiopathic oligozoospermia undergone Prospective, randomized, double-blind, placebo-controlled clinical study. | Significant improvement in both morphology and forward motility at the fifth month, total sperm count, sperm concentration, forward motility and morphology were also improved. | Available,FDA approved for spermatogenesis induction(Treatment of oligozoospermia) | |
300 IU, Subcutaneously | Every other day for ≥ 4 months | A randomized double-blind placebo controlled study on seminal parameter in 30 normogonadotrophic patients | Improved sperm concentration and total sperm number | Available, FDA approved for treatment of infertility due to gonadotropin deficiency | (Dabaja & Schlegel, 2014), (Paradisi, Busacchi, Seracchioli, Porcu, & Venturoli, 2006) |
The majority of side effects are minor and unusual (<10%), such as libido changes, edema, breast soreness, headaches or asthenia without overlooking about impaired mineralization and augmented bone resorption in prolonged therapies (García-Baquero et al., 2020).
Hormonal therapy
The major objective of GnRH (Gonadotropin releasing hormone) treatment is promoting gonadotropin release from the anterior pituitary and subsequent pathways in the HPG (Hypothalamic pituitary gonadal axis) Dabaja and Schlegel (2014). Rendering to dataset sorted from newly published meta-analysis, empirical gonadotropin treatment boosts the total sperm count and augments live birth rates and pregnancy rates (García-Baquero et al., 2020).
According to the Cochrane database analysis, 456 members(with confirmed treatment and follow-up studies) undergone six randomised controlled trials demonstrated an increase in conception and live birth rates upon 3 months of randomly assigned GnRH treatment (Attia, Abou-Setta, & Hg, 2013).Gynecomastia, increased breast soreness, and other side effects like local injection site irritation were uncommon and moderate in this instance (Dabaja & Schlegel, 2014). A clinical study using recombinant follicle stimulating hormone given alternatively with a dose of 300 IU througout 5 months on 365 men with idiopathic oligozoospermia was conducted in a double-blinded, prospective, randomized, placebo-controlled fashion. It further prompted the spermatogenetic process thereby result in substantial enhancement of semen parameters (Ding et al., 2015). Male infertility brought on by disorders of the hypothalamus responds well to pulsatile gonadotropin releasing hormone (GnRH), whereas male infertility brought on by pituitary insufficiency is typically managed with human chorionic gonadotropin (hCG) in combination with human menopausal gonadotropin (hMG), purified urinary FSH or recombinant human FSH (Mohamed et al., 2020).
Anti-estrogens
Based on a recent meta-analysis, empirical antiestrogen therapy (clomiphene 50 mg/24 - 48 h, tamoxifen 20 mg/24 h) elevates the frequencies of conception, pregnancy, and sperm motility rates (Dabaja & Schlegel, 2014). A comprehensive analysis, together with the meta-analysis of prospective studies and RCTs were undertaken based on the effect of Clomiphene Citrate on concentration of sperms. Data from one prospective study and two randomized controlled trials on 197 men suffering from oligozoospermia showed significant increase in sperms of nearly 7.7 million sperms (Bridges et al., 2015). A substantial rise in pregnancy rate was seen in a double-blinded, controlled study where the patients were allocated randomly to receive either a placebo or 100 mg/day of CC (Clomiphene Citrate) for 10 days each month for three months. This suggested that CC is an effective treatment (Sokol, Steiner, Bustillo, Petersen, & Swerdloff, 1988). The comparison of effectiveness of treatment using a clomiphene and vitamin E combination to a placebo in a different group of 60 subfertile males was studied. Here the infertile patients were divided into 2 groups and treated with either a placebo (n=30) or a combination of clomiphene citrate (25 mg/day) and vitamin E (400 mg/day) for 6 months. When compared to the placebo treated group, the treatment group exhibited a notable improvement in spermatozoal concentration and forward motility (Puia & Pricop, 2022).
CONCLUSION
Conferring to World Health Organization guidelines (Schlatt & Behre, 2010), conventional semen analysis is a crucial diagnostic and therapeutic technique for treating male fecundity problems. It primarily considered as descriptive microscopic analysis of sperm mobility, concentration and morphology and aids in (i) recognising a specific sperm disorders, (ii) predicting the outcomes of assisted reproductive technology (ART), (iii) assessing the negative effects of environmental, dietary and chemical pollutants and (iv) directing clinicians toward particular diagnostic and therapeutic approaches (Magdi et al., 2017).
The empirical therapy of male idiopathic infertility is offered in a wide range of medicinal modalities. Our objective is to offer these group a therapeutic options that are secure, efficient and may even eliminate needless ART, as it is clear that a healthy diet and careful supplements may improve the rate of conception by reducing the effect of male infertility. According to the review, antioxidants have a clear impact on some semen parameters notably comprising sperm motility, count and morphology based on various preclinical research. So for the purpose of reversing OS-induced sperm malfunction and to increase the conception rates, antioxidants were generally found to have a positive effect. Studies conducted preclinically have shown that a variety of antioxidant aspects provide a positive impact on pregnancy rates, sperm concentration and live birth rates.
The efficiency of the plant extract and/or its medicine preparation must be evaluated in human patients following a sequence of standardised safety studies in animal models. Previous RCTs (Randomized clinical trials) have emphasized how medicinal plants can increase conception rates. Further randomized trials are essential to bring the aforementioned effect and establish the maximum dosage and extent of treatment with herbal remedies in order to create a novel strategy for treating male infertility. The advancement of novel botanical medications for the treatment of male infertility is anticipated to be sparked by these intriguing discoveries.
In addition to all of these, it is worth mentioning that a wide range of medicinal therapies are better to be offered for the first-hand treatment of male idiopathic OATs. All of the substances we've discussed, whether it is a hormonal or not, tends to have the potential in managing male infertility. In order to prevent predictable therapeutic failures, it is vital to select the best suitable treatment options based on the clinical features of every patient. In conclusion, the analysis of the available evidence, which is based on a number of randomized controlled trials, reveals that the practice of synthetic, herbal drugs, as well as oral antioxidant supplements, may help men who really experiences difficulties in conceiving, particularly by enhancing certain type of semen parameters.